1. What is the projected Compound Annual Growth Rate (CAGR) of the Life Science CDMO?
The projected CAGR is approximately 6.41%.
 Life Science CDMO
Life Science CDMOLife Science CDMO by Application (Pharmaceutical, Clinical, Research), by Type (Active Pharmaceutical Ingredient (API) CDMO, Biologics CDMO, Finished Dosage Form (FDF) CDMO, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The Life Science Contract Development and Manufacturing Organization (CDMO) market is experiencing robust growth, driven by the increasing outsourcing of drug development and manufacturing by pharmaceutical and biotechnology companies. This trend is fueled by several factors, including the rising complexity of drug development, the need for specialized expertise and technologies (particularly in biologics), and the desire for greater efficiency and cost-effectiveness. The market is segmented by application (pharmaceutical, clinical, research) and by type of service (API CDMO, Biologics CDMO, FDF CDMO, and others). Biologics CDMO is a particularly high-growth segment, reflecting the increasing prevalence of biologics in the pharmaceutical pipeline. The geographically diverse market sees strong performance in North America and Europe, driven by established pharmaceutical industries and robust regulatory frameworks. However, Asia-Pacific is emerging as a significant growth region, fueled by increasing investments in R&D and manufacturing infrastructure, particularly in China and India. Challenges include stringent regulatory requirements, fluctuating raw material costs, and competition among established players and emerging CDMOs.
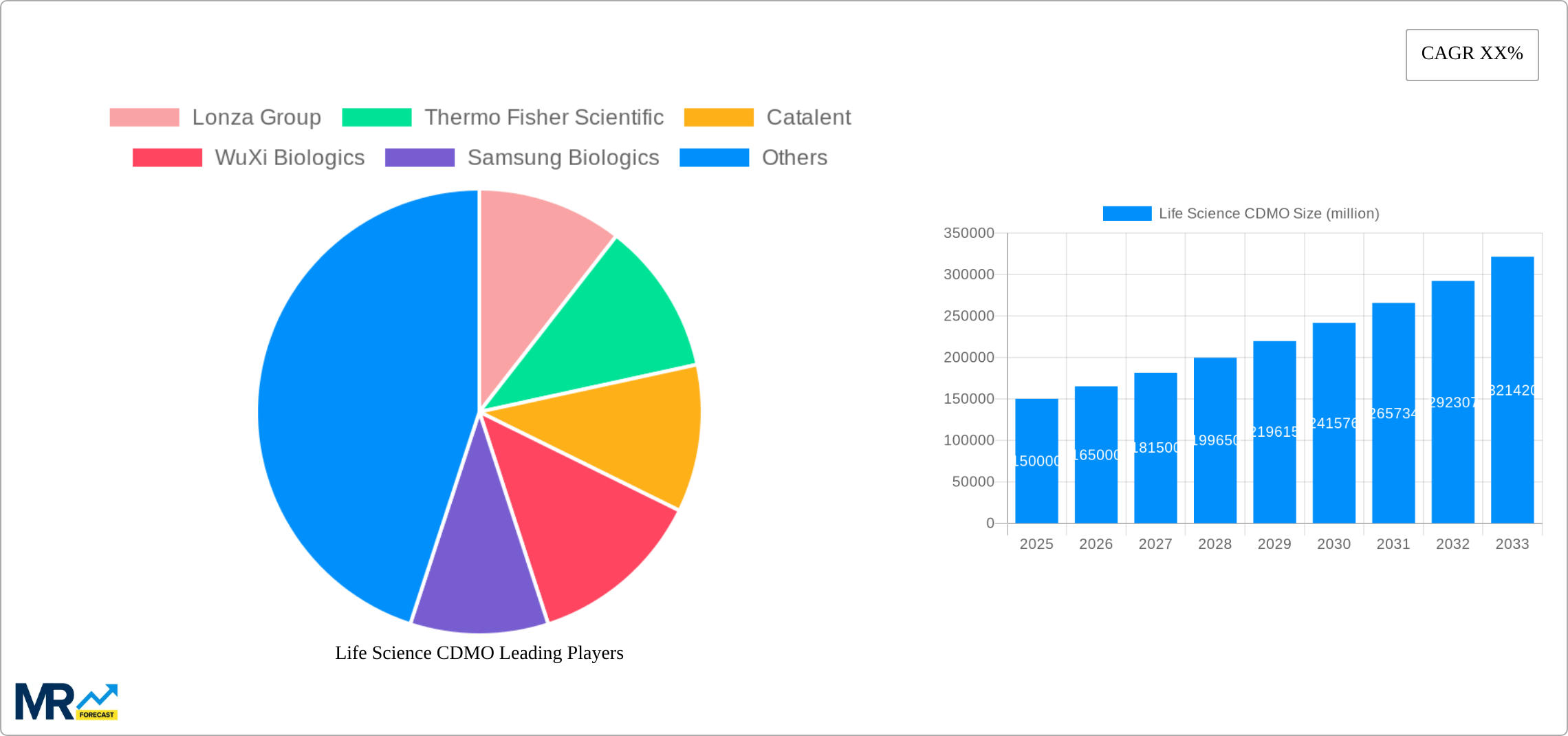

The forecast period (2025-2033) anticipates continued expansion, with a projected CAGR (Compound Annual Growth Rate) that reflects a dynamic market. While the exact CAGR is not provided, a reasonable estimate considering the drivers and trends described above could be within the range of 8-12%, reflecting strong but sustainable growth. This growth will be driven by continuous innovation in drug development, the increasing adoption of advanced manufacturing technologies, and the expanding pipeline of novel therapies. The market's competitiveness will intensify as new players enter the market, while existing players consolidate and expand their service offerings through acquisitions and strategic partnerships. Future growth will depend on the ability of CDMOs to adapt to evolving regulatory landscapes, provide innovative solutions, and meet the demands of an increasingly complex and demanding pharmaceutical industry.
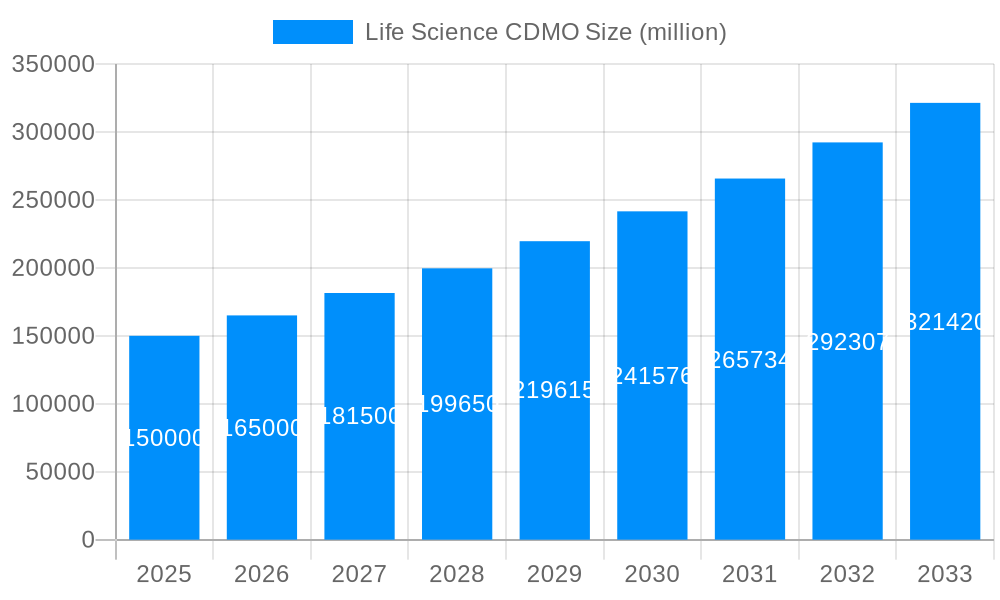

The life science contract development and manufacturing organization (CDMO) market is experiencing robust growth, projected to reach several billion USD by 2033. This expansion is driven by several converging factors, including the increasing outsourcing of drug development and manufacturing activities by pharmaceutical and biotechnology companies. The rising complexity of drug development, particularly in the biologics space, necessitates specialized expertise and infrastructure, which many companies find more cost-effective to outsource. This trend is further fueled by the burgeoning pipeline of novel therapeutics, including cell and gene therapies, which demand highly specialized CDMO services. The market is witnessing a consolidation trend, with larger CDMOs acquiring smaller companies to expand their service offerings and geographic reach. This consolidation is improving efficiency and economies of scale. Furthermore, technological advancements in drug development and manufacturing, such as automation and process intensification, are enhancing productivity and reducing production costs, which positively impacts overall market growth. The increasing focus on personalized medicine and advanced therapies also creates significant opportunities for CDMOs specializing in these areas. Finally, the regulatory landscape plays a role; stringent regulatory requirements for drug safety and efficacy necessitate significant investment in quality control and compliance, driving demand for CDMOs with robust quality management systems. Overall, the life science CDMO market presents a dynamic and promising landscape with continued expansion across various segments and geographies.
Several key factors are propelling the significant growth of the life science CDMO market. Firstly, the escalating cost of in-house drug development and manufacturing is pushing pharmaceutical and biotechnology companies to strategically outsource these functions. This allows companies to focus on core competencies like research and development and commercialization while leveraging the expertise and infrastructure of specialized CDMOs. Secondly, the complexity of modern drug development, particularly in areas like biologics, cell and gene therapies, requires advanced technologies and specialized expertise beyond the capabilities of many smaller or mid-sized pharmaceutical companies. CDMOs offer access to cutting-edge technologies and experienced professionals, facilitating faster and more efficient drug development. The increasing demand for faster drug development timelines and time-to-market pressures further incentivize outsourcing. CDMOs, with their established facilities and processes, can often deliver faster turnaround times compared to in-house development, allowing for quicker product launches. Finally, stringent regulatory compliance necessitates substantial investment in quality control and validation. CDMOs often possess pre-approved facilities and robust quality management systems, reducing the regulatory burden for their clients and offering them peace of mind.
Despite the significant growth potential, the life science CDMO market faces certain challenges and restraints. Maintaining consistent quality and compliance across diverse projects and geographical locations remains a significant hurdle for many CDMOs. Meeting stringent regulatory requirements worldwide, especially for complex therapies like cell and gene therapies, requires robust quality management systems and meticulous documentation. Furthermore, the increasing demand for specialized CDMO services can lead to capacity constraints, resulting in longer lead times and potential delays in project timelines. Competition is intense within the CDMO market, with established players and emerging companies vying for market share. Managing supply chain risks is critical, especially considering the global nature of this industry. Disruptions in raw material supply or transportation can significantly impact manufacturing schedules and timelines. The need for continuous investment in technology upgrades and personnel training to maintain competitiveness and adapt to evolving industry trends presents a significant financial burden for CDMOs. Finally, intellectual property (IP) protection is a significant concern. Ensuring confidentiality and safeguarding clients' valuable intellectual property throughout the development and manufacturing process is crucial for maintaining trust and building strong client relationships.
The Biologics CDMO segment is poised for significant growth and is expected to dominate the market during the forecast period (2025-2033). This is driven by the increasing prevalence of biologic therapies, including monoclonal antibodies, recombinant proteins, and cell and gene therapies, which command higher prices and exhibit impressive growth trajectories. Several key regions are contributing significantly to this growth.
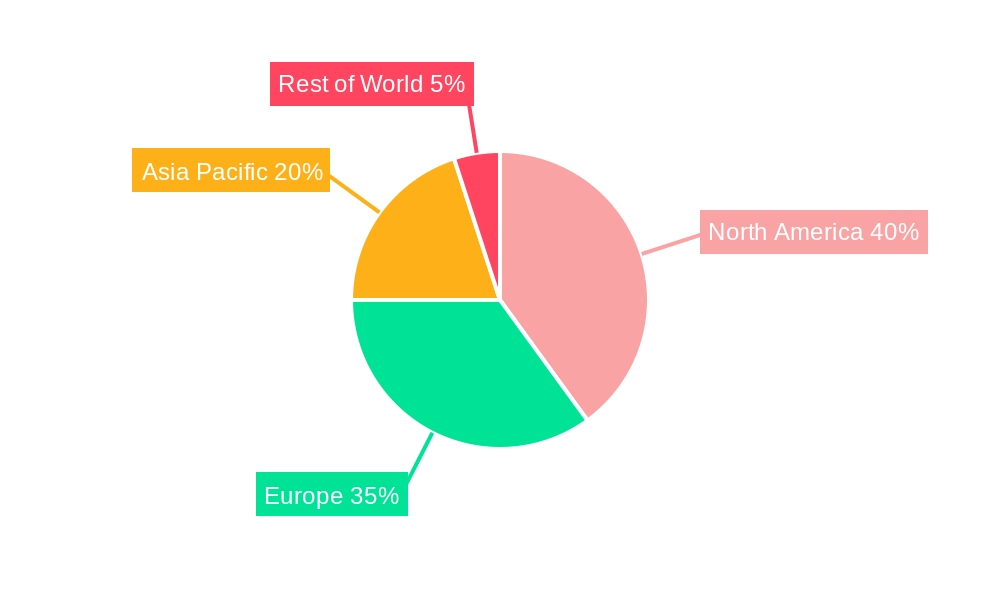
North America: The US, in particular, boasts a strong presence of major pharmaceutical companies, advanced research infrastructure, and a highly developed regulatory framework, making it a prime market for biologics CDMO services. Investment in facilities and advanced technologies is high. The market value is projected to exceed $XXX million by 2033.
Europe: Countries like Germany, the UK, and Switzerland have established themselves as hubs for pharmaceutical manufacturing and possess a strong regulatory environment and skilled workforce, driving significant growth in this region. The market value is estimated at over $XXX million by 2033.
Asia Pacific: This region is experiencing rapid growth in the biopharmaceutical sector. China, India, Japan, and South Korea are emerging as major players, with increasing investments in CDMO facilities and growing demand for biologics. The market is projected to exceed $XXX million by 2033.
Within these regions, growth is further fueled by:
The life science CDMO industry's growth is fueled by increasing outsourcing trends, the rising complexity of drug development requiring specialized expertise, advancements in technology such as automation and process intensification, and the burgeoning demand for personalized medicines. These factors together create a synergistic effect, driving substantial growth and innovation within the sector.
This report provides a comprehensive analysis of the life science CDMO market, encompassing market size, growth drivers, challenges, key players, and significant developments. The detailed segmentation of the market by application (pharmaceutical, clinical, research), type of CDMO service (API, biologics, FDF), and geographical region offers a granular view of the market dynamics. The report also includes detailed financial projections and forecasts, enabling stakeholders to make informed strategic decisions. The combination of quantitative and qualitative insights provides a holistic perspective on the opportunities and challenges facing the life science CDMO industry.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.41% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 6.41%.
Key companies in the market include Lonza Group, Thermo Fisher Scientific, Catalent, WuXi Biologics, Samsung Biologics, Siegfried, Fujifilm Diosynth Biotechnologies, Lars Petersen, Recipharm, Boehringer Ingelheim, MilliporeSigma, Aenova Group, AGC Parma Chemicals, GenScript, Novartis, Sai Life Sciences, .
The market segments include Application, Type.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A.
Yes, the market keyword associated with the report is "Life Science CDMO," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Life Science CDMO, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.