1. What is the projected Compound Annual Growth Rate (CAGR) of the FDA Verified Service?
The projected CAGR is approximately XX%.
 FDA Verified Service
FDA Verified ServiceFDA Verified Service by Type (Food Factory, Radiation Source Laser Class, Medical Machinery), by Application (Personal, Commercial), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The FDA Verified Service market is experiencing robust growth, driven by increasing demand for ensuring product safety and compliance across diverse sectors. The market's expansion is fueled by stricter regulatory environments globally, particularly within the food and medical device industries. Companies are prioritizing FDA verification to enhance consumer trust, avoid costly recalls, and maintain a competitive edge. The rising adoption of advanced technologies for testing and verification, such as laser-based systems, further contributes to market growth. While precise market sizing data is unavailable, considering a plausible CAGR (let's assume 8% based on industry averages for similar regulated markets) and a 2025 market value in the range of $250 million, we can project significant expansion over the forecast period (2025-2033). The Food Factory and Medical Machinery segments are key drivers, with substantial demand from both personal and commercial applications. Geographical expansion is also anticipated, with North America and Europe currently holding significant market shares, while Asia-Pacific is poised for considerable growth due to rising industrialization and stringent regulatory reforms. However, high verification costs and a complex regulatory landscape could pose challenges to market expansion. This calls for streamlined processes and innovative solutions that make FDA verification more accessible and cost-effective for smaller companies. Furthermore, consistent education and awareness regarding the importance of FDA verification will be crucial to maintain market momentum.
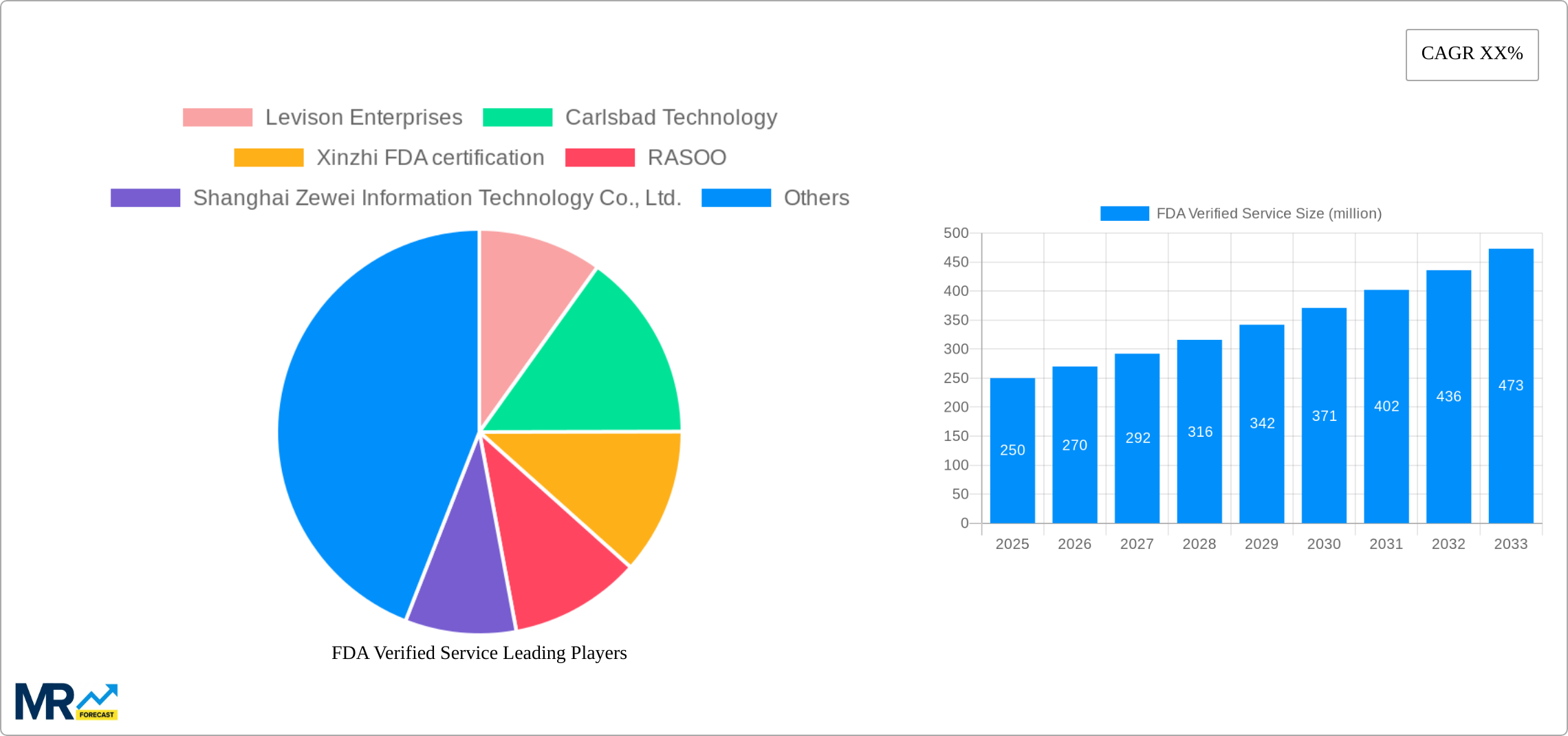

The competitive landscape is fragmented, with numerous companies offering FDA verification services across various regions. The presence of both large multinational corporations and smaller, specialized firms indicates a diverse range of service offerings and pricing strategies. The focus for companies operating in this market should be on specializing in specific niches within food and medical industries and developing specialized technologies that provide faster and more efficient verification solutions. Geographic expansion and strategic partnerships with regulatory bodies can enhance market penetration and sustainability. By addressing the challenges of cost and complexity, the FDA Verified Service market is positioned for continued growth driven by both expanding regulatory requirements and technological innovation.
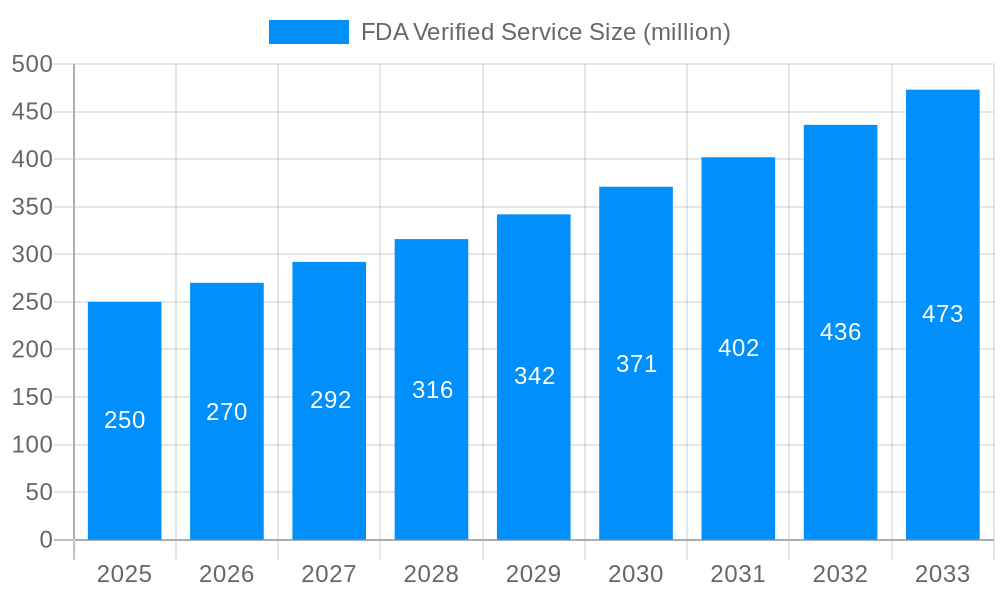

The FDA Verified Service market is experiencing robust growth, projected to reach multi-million unit volumes by 2033. Analysis from the period 2019-2024 (Historical Period) reveals a steady upward trajectory, with the base year 2025 marking a significant inflection point. The estimated market size for 2025 signals substantial expansion, driven by increasing global demand for FDA-compliant products across diverse sectors. This demand is further amplified by stringent regulatory requirements and the rising consumer awareness of product safety and quality. The forecast period (2025-2033) anticipates continued, albeit potentially fluctuating, growth fueled by technological advancements within the FDA verification process itself, making it more efficient and accessible to a wider range of businesses. Key market insights highlight a strong correlation between increased global trade and the need for FDA verification services, particularly within the medical machinery and food processing industries. The market’s expansion isn't solely dependent on the number of products needing verification but also on the evolving complexity of regulatory compliance, leading businesses to seek specialized and reliable third-party verification services. This trend is likely to persist, offering promising opportunities for growth within the FDA Verified Service sector in the coming years. The study period of 2019-2033 provides a comprehensive understanding of market evolution, factoring in both past performance and future projections.
Several key factors are driving the growth of the FDA Verified Service market. Firstly, the increasing stringency of FDA regulations across various industries, particularly concerning food safety and medical device compliance, compels manufacturers to proactively seek verification services. This is not merely a matter of regulatory compliance; it's also a significant factor in maintaining brand reputation and securing consumer trust. Secondly, the globalization of trade necessitates compliance with international standards, making FDA verification a crucial aspect of exporting products to the US market and other regions with similar regulatory frameworks. Thirdly, the rising complexity of product development and manufacturing processes increases the likelihood of errors and necessitates the expertise of specialized service providers to ensure regulatory conformity. Furthermore, the growing adoption of advanced technologies within FDA verification processes, such as automated testing and data analysis, is improving efficiency and reducing costs, making the service more accessible to a wider range of companies. Finally, the rising consumer awareness of product safety and quality, coupled with increased transparency, places greater pressure on businesses to demonstrate compliance through independent verification. These combined forces create a powerful impetus for the continued expansion of the FDA Verified Service market.
Despite the significant growth potential, the FDA Verified Service market faces several challenges and restraints. The primary challenge is the high cost associated with obtaining FDA verification, which can be a significant barrier, particularly for small and medium-sized enterprises (SMEs). This cost factor often involves not only the fees for the service itself but also the potential need for product redesign or reformulation to meet regulatory standards. Furthermore, the evolving nature of FDA regulations presents an ongoing challenge, requiring service providers to constantly adapt and update their knowledge and procedures to maintain compliance. The complexity of the regulatory landscape can also lead to lengthy verification processes, potentially delaying product launches and impacting market competitiveness. Another key restraint is the potential for variability in the quality and reliability of service providers, demanding meticulous due diligence from companies selecting a verification partner. Finally, geopolitical uncertainties and disruptions to global supply chains can indirectly affect the market by impacting production timelines and the availability of resources required for compliance.
The Medical Machinery segment is expected to dominate the FDA Verified Service market due to the stringent regulatory requirements governing medical devices. The inherent risk associated with faulty medical equipment necessitates rigorous testing and verification, fueling demand for specialized FDA verification services. This is further compounded by increasing innovation and technological advancements within the medical device industry, leading to more complex products requiring specialized verification expertise.
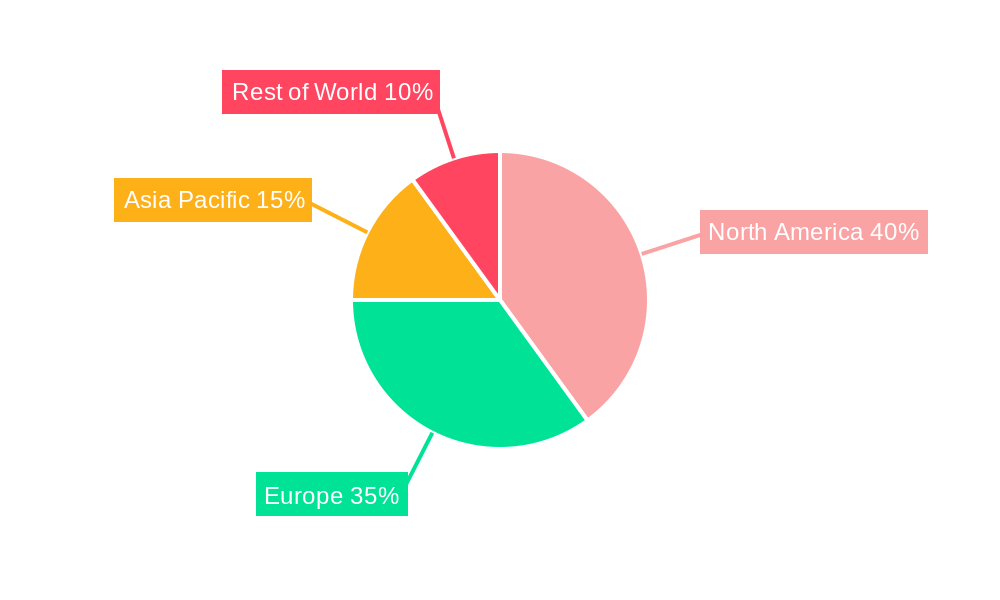
High Growth Potential in North America: The United States, as the origin of the FDA, is a pivotal market, driving the significant demand for FDA verification services. The robust regulatory framework and the strong emphasis on consumer safety solidify this region's leading position.
Asia-Pacific's Emerging Market: While North America currently holds the top spot, the Asia-Pacific region presents a significant opportunity for growth. The region's expanding medical device manufacturing sector and increasing regulatory scrutiny are key factors contributing to this potential. This growth is likely to be driven by increasing disposable incomes, improvements in healthcare infrastructure, and a rising demand for high-quality medical equipment.
European Union's Stringent Regulations: The European Union, with its own robust regulatory framework for medical devices, presents another key market. Although perhaps not as directly tied to FDA verification, companies often leverage overlapping compliance elements to streamline their processes.
In summary, the convergence of stringent regulations, growing product complexity, and increasing consumer awareness across the medical machinery sector paints a picture of sustained growth for FDA verification services, particularly in North America and with high potential in the Asia-Pacific region.
The increasing global demand for FDA-compliant products, combined with stricter regulatory enforcement and advancements in verification technologies, are primary growth catalysts. The growing awareness among businesses of the importance of product safety and quality, coupled with the potential negative consequences of non-compliance, drives them to seek professional FDA verification services. This, along with the simplification of certain verification procedures, makes the services more accessible and cost-effective.
This report offers a detailed analysis of the FDA Verified Service market, covering historical data, current market dynamics, and future projections. It provides valuable insights into market trends, growth drivers, challenges, and key players, enabling informed decision-making for businesses and investors alike. The comprehensive nature of the report allows stakeholders to assess risks and opportunities effectively, positioning themselves strategically within this rapidly evolving market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XX% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately XX%.
Key companies in the market include Levison Enterprises, Carlsbad Technology, Xinzhi FDA certification, RASOO, Shanghai Zewei Information Technology Co., Ltd., Guangzhou Anpu Testing Technology Service Co., Ltd., Dongguan Xinzhun Testing Technology Service Co., Ltd., Shenzhen Shangtong Testing Technology Co., Ltd., Shenzhen Xunke Commodity Inspection Co., Ltd., Zhejiang Sunrise International Engineering Design Co., Ltd., .
The market segments include Type, Application.
The market size is estimated to be USD XXX million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million.
Yes, the market keyword associated with the report is "FDA Verified Service," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the FDA Verified Service, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.