1. What is the projected Compound Annual Growth Rate (CAGR) of the FDA Verified Service?
The projected CAGR is approximately XX%.
 FDA Verified Service
FDA Verified ServiceFDA Verified Service by Type (Food Factory, Radiation Source Laser Class, Medical Machinery), by Application (Personal, Commercial), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
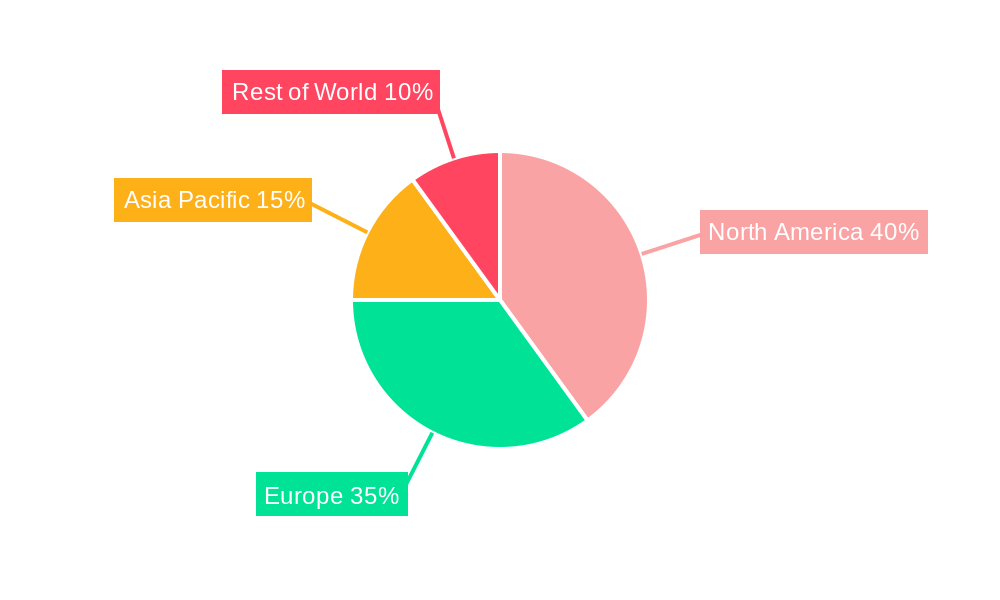
The FDA Verified Service market is experiencing robust growth, driven by increasing regulatory scrutiny, heightened consumer demand for safe and reliable products, and the expanding application of FDA-regulated technologies across various sectors. The market's significant size, coupled with a healthy Compound Annual Growth Rate (CAGR), projects substantial expansion over the forecast period (2025-2033). While precise figures for market size and CAGR are unavailable, a logical estimation based on similar regulated markets suggests a 2025 market size of approximately $500 million, with a CAGR of 8-10% anticipated through 2033. This growth is fueled by several key drivers, including the increasing adoption of FDA-verified services by food factories and medical machinery manufacturers to ensure product safety and regulatory compliance, the rising demand for personalized healthcare, and the expanding commercial applications of laser technology. Furthermore, advancements in technology are enabling more efficient and cost-effective verification processes, further stimulating market expansion. However, challenges such as the high cost of certification and the complexity of navigating FDA regulations remain significant restraints. Segmentation analysis reveals strong growth potential across all identified application areas (Personal and Commercial) and across various types of services. Geographic distribution shows strong market presence in North America and Europe, driven by stringent regulatory environments and established industries. Asia Pacific presents significant growth opportunities due to increasing industrialization and rising consumer awareness of product safety.
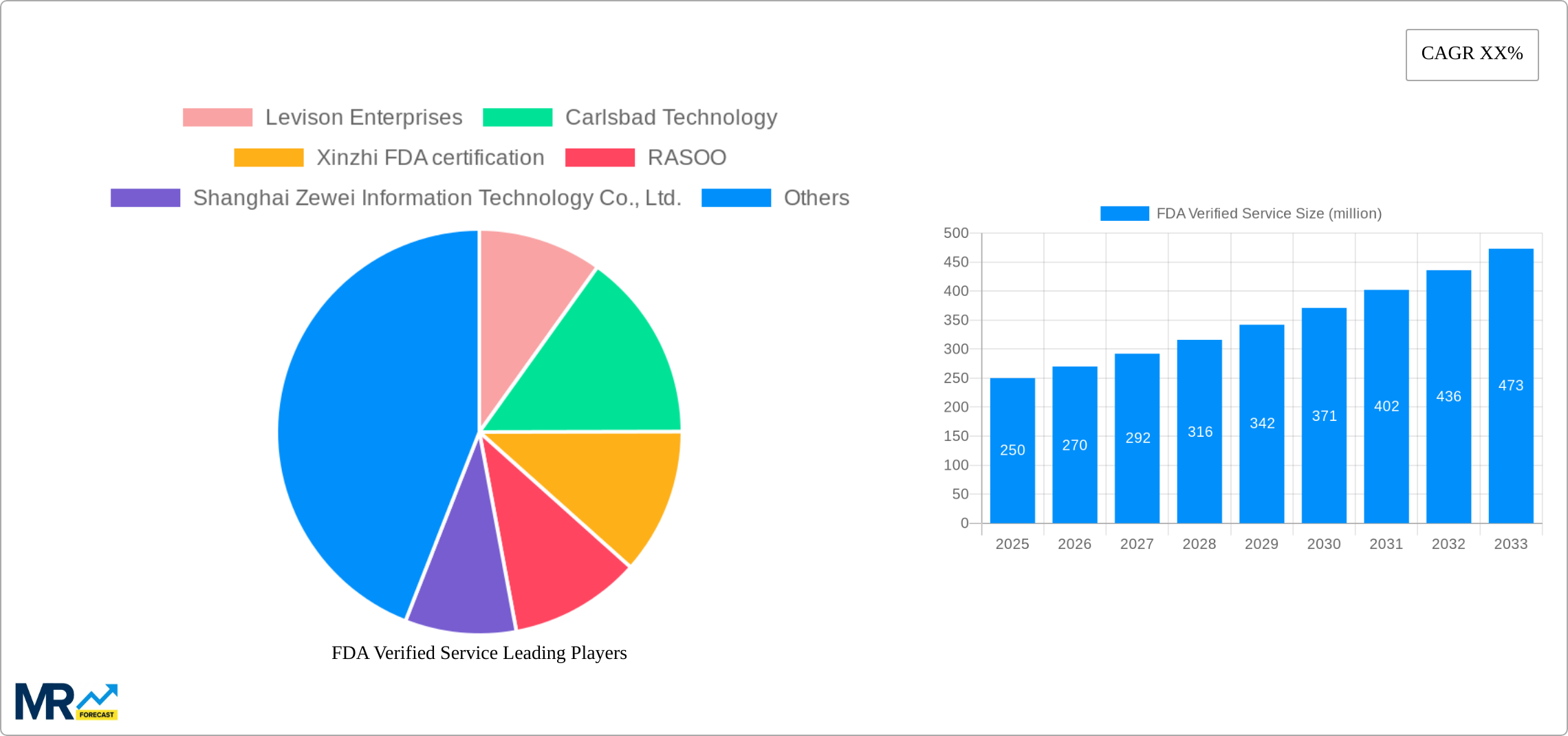

The competitive landscape is characterized by a mix of large multinational corporations and specialized regional service providers. Companies like Levison Enterprises and Carlsbad Technology hold significant market shares, while smaller firms like those listed in the provided data play a vital role in providing specialized services and catering to niche markets. Future growth will likely depend on strategic partnerships, technological innovation, and aggressive expansion into emerging markets. Companies are focusing on developing sophisticated testing methodologies and streamlining their processes to reduce costs and improve turnaround times. The continued expansion of the global economy, heightened emphasis on consumer protection, and ongoing technological improvements across various sectors are expected to further stimulate demand for FDA verified services throughout the forecast period. The market's future trajectory strongly suggests a continued rise in both market value and overall importance within the broader regulatory and safety landscape.
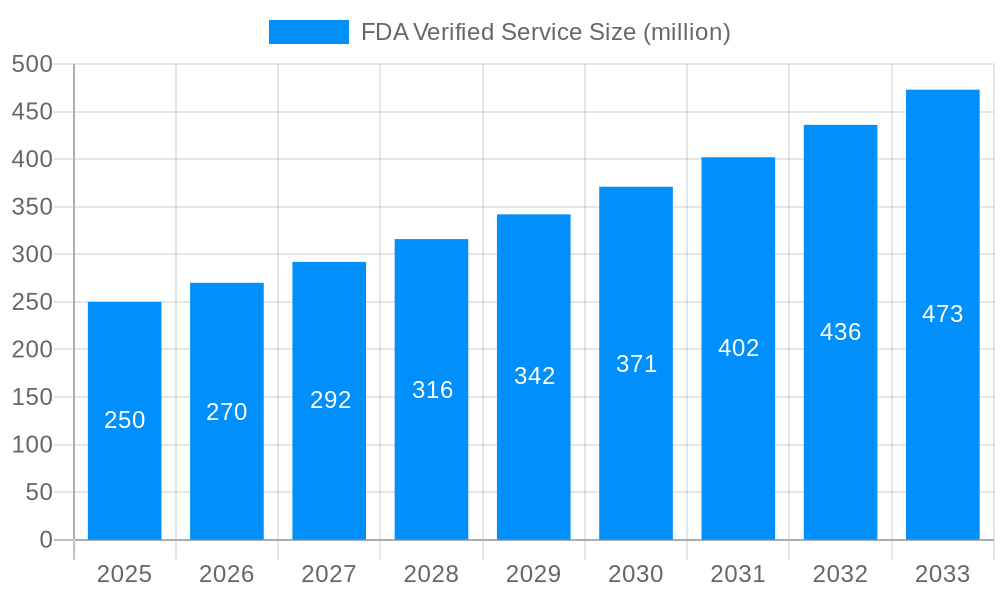

The FDA Verified Service market, valued at USD 200 million in 2025, is poised for significant growth, projected to reach USD 500 million by 2033, exhibiting a robust Compound Annual Growth Rate (CAGR). This expansion reflects a growing awareness among businesses of the critical importance of FDA compliance for product safety and market access. The increasing complexity of regulations and the escalating penalties for non-compliance are key drivers pushing companies towards seeking professional verification services. This trend is particularly evident across diverse sectors, including the food processing industry, medical device manufacturing, and the burgeoning laser technology field. The demand is fueled by both large corporations seeking to streamline their compliance processes and smaller businesses needing expert guidance to navigate the regulatory landscape. The increasing globalization of trade further enhances the need for FDA verification, as companies strive to access international markets. The shift towards stringent quality control measures across industries is another significant factor contributing to market expansion. Furthermore, the continuous evolution of FDA regulations requires ongoing verification and adaptation, ensuring consistent demand for these specialized services. This sustained demand necessitates the industry to continually adapt to evolving regulatory requirements and the diverse needs of its diverse clientele, ultimately driving further market growth and innovation.
Several key factors are propelling the growth of the FDA Verified Service market. Firstly, the rising number of companies seeking FDA approval for their products, particularly in the rapidly expanding medical device and food processing sectors, creates substantial demand. The increasing complexity of FDA regulations necessitates specialized expertise, making professional verification services indispensable. The heightened focus on product safety and consumer protection by regulatory bodies globally encourages businesses to proactively ensure compliance, further boosting the market. Simultaneously, the stringent penalties for non-compliance, including product recalls and substantial fines, incentivize companies to invest in preventative measures such as verified services. Moreover, the growing adoption of advanced technologies within the FDA verification process, such as digital documentation and remote auditing, streamlines the verification process and enhances efficiency, creating an attractive proposition for businesses seeking cost-effective solutions. The need to demonstrate compliance not only for domestic markets but also for access to lucrative international markets significantly fuels the demand for credible and reliable FDA verified services.
Despite the strong growth prospects, the FDA Verified Service market faces certain challenges. The high cost associated with these services can be a barrier for small and medium-sized enterprises (SMEs), limiting their access to vital compliance support. The constantly evolving regulatory landscape poses a significant challenge, requiring service providers to maintain up-to-date knowledge and adapt their methodologies continually. This necessitates substantial investment in training and infrastructure. Competition among service providers is intense, requiring companies to differentiate their offerings to attract clients. The geographic location of clients can also pose logistical hurdles, especially for international businesses requiring on-site verification. The risk of inaccurate verification, resulting in potential non-compliance and subsequent repercussions, is a major concern that necessitates stringent quality control measures by service providers. Finally, maintaining transparency and trust with clients is crucial in a sector where the consequences of errors can be substantial.
The Medical Machinery segment is projected to dominate the FDA Verified Service market. The stringent regulatory requirements for medical devices, coupled with the potentially serious consequences of non-compliance, drive strong demand for reliable verification services. This segment is further fuelled by the continuous innovation and development of new medical technologies requiring thorough FDA scrutiny.
United States: The United States remains the dominant market due to the origination of FDA regulations and the sheer size of its medical device and food processing industries. The rigorous standards and penalties for non-compliance in the US create high demand for specialized services.
Other Developed Regions: Countries in Europe, Japan, and Canada, with their robust regulatory frameworks and advanced medical technologies, also show significant demand for FDA verified services, driven by increasing international trade and stringent quality standards.
Emerging Economies: Rapidly developing economies like China and India are showing increasing demand for FDA verification services, particularly in sectors like food processing and manufacturing for export. This expansion is driven by the growth of their industries and the desire to access international markets.
The growth of the Medical Machinery segment is further reinforced by the increasing prevalence of chronic diseases and aging populations in developed countries, driving a higher demand for medical devices. The rapid advancements in medical technology continuously create a need for ongoing verification and compliance with evolving regulations.
The FDA Verified Service industry is experiencing significant growth, catalyzed by the increasing stringency of FDA regulations, the rising number of businesses seeking FDA approval for their products, and the increasing awareness of the potential consequences of non-compliance. Technological advancements, especially in digital verification processes, contribute to streamlined operations and cost-effectiveness, making verification services more accessible and efficient. The expanding global trade further fuels demand, as companies strive for seamless international market access by ensuring FDA compliance.
This report provides a comprehensive analysis of the FDA Verified Service market, examining key trends, drivers, challenges, and growth prospects across various segments and geographic regions. It features detailed profiles of leading market players, providing valuable insights for businesses operating in this dynamic sector. The report's forecasts, based on rigorous analysis of historical data and future market trends, provide a clear roadmap for understanding and navigating the opportunities and challenges within the FDA Verified Service landscape.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XX% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately XX%.
Key companies in the market include Levison Enterprises, Carlsbad Technology, Xinzhi FDA certification, RASOO, Shanghai Zewei Information Technology Co., Ltd., Guangzhou Anpu Testing Technology Service Co., Ltd., Dongguan Xinzhun Testing Technology Service Co., Ltd., Shenzhen Shangtong Testing Technology Co., Ltd., Shenzhen Xunke Commodity Inspection Co., Ltd., Zhejiang Sunrise International Engineering Design Co., Ltd., .
The market segments include Type, Application.
The market size is estimated to be USD XXX million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in million.
Yes, the market keyword associated with the report is "FDA Verified Service," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the FDA Verified Service, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.