1. What is the projected Compound Annual Growth Rate (CAGR) of the Drug Repositioning?
The projected CAGR is approximately 4.5%.
 Drug Repositioning
Drug RepositioningDrug Repositioning by Type (Generic Drugs, Clinical Trial Failed Drugs), by Application (Cardiovascular Diseases, Mental Illness, Cancer, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The drug repositioning market, valued at $27.03 billion in 2025, is projected to experience robust growth, driven by several key factors. The increasing prevalence of chronic diseases like cardiovascular diseases, mental illnesses, and cancer, coupled with the high cost of developing novel drugs, fuels the demand for repurposing existing medications. This approach significantly reduces the time and cost associated with drug development, offering a faster and more cost-effective pathway to market for pharmaceutical companies. Furthermore, advancements in genomics, bioinformatics, and artificial intelligence are accelerating the identification of potential drug repositioning candidates and enhancing the precision of clinical trials, leading to improved success rates. The market is segmented by drug type (generic drugs and clinical trial failed drugs) and therapeutic application (cardiovascular diseases, mental illness, cancer, and others), with each segment demonstrating unique growth trajectories influenced by specific disease prevalence, regulatory landscapes, and research priorities. Competition in the market is fierce, with major pharmaceutical players like Teva, Novartis, Mylan, Johnson & Johnson, Pfizer, and others actively engaged in drug repurposing initiatives. This competitive landscape fosters innovation and accelerates the identification and development of new therapeutic uses for existing drugs.
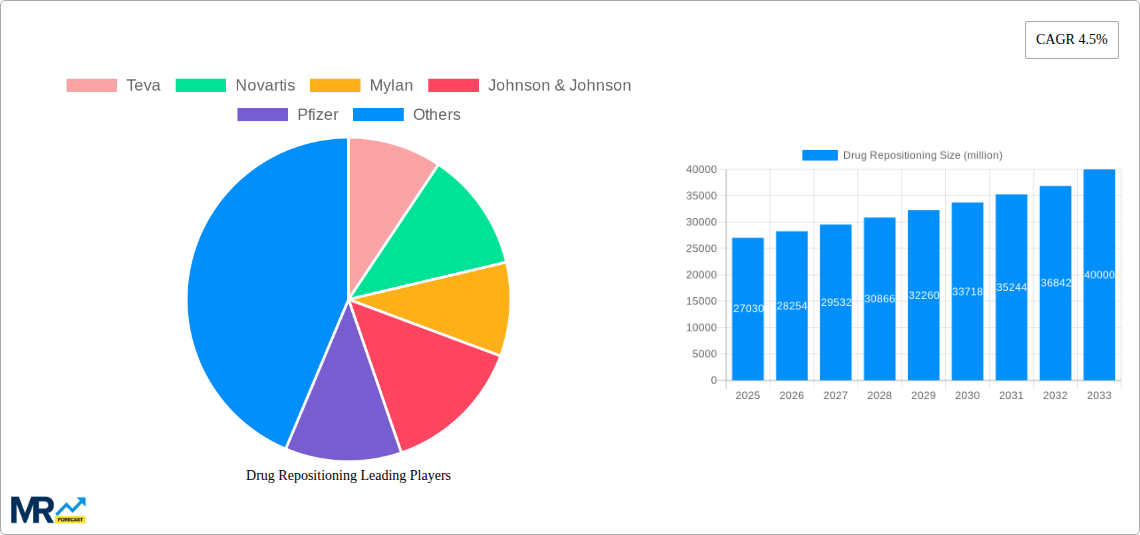

The geographical distribution of the drug repositioning market is geographically diverse, with North America and Europe currently holding significant market shares due to robust healthcare infrastructure, advanced research capabilities, and stringent regulatory frameworks. However, the Asia-Pacific region, particularly India and China, is expected to witness substantial growth in the coming years, driven by rising healthcare expenditure, increasing disease burden, and growing pharmaceutical manufacturing capabilities. Government initiatives and funding for research and development in drug repositioning are further catalyzing market expansion in these regions. The consistent CAGR of 4.5% indicates a steady and sustained growth trajectory for the forecast period (2025-2033), underpinned by the ongoing need for affordable and effective treatment options for a wide range of diseases. Successful drug repositioning projects contribute to improved patient outcomes, reduced healthcare costs, and a strengthened pharmaceutical innovation pipeline.
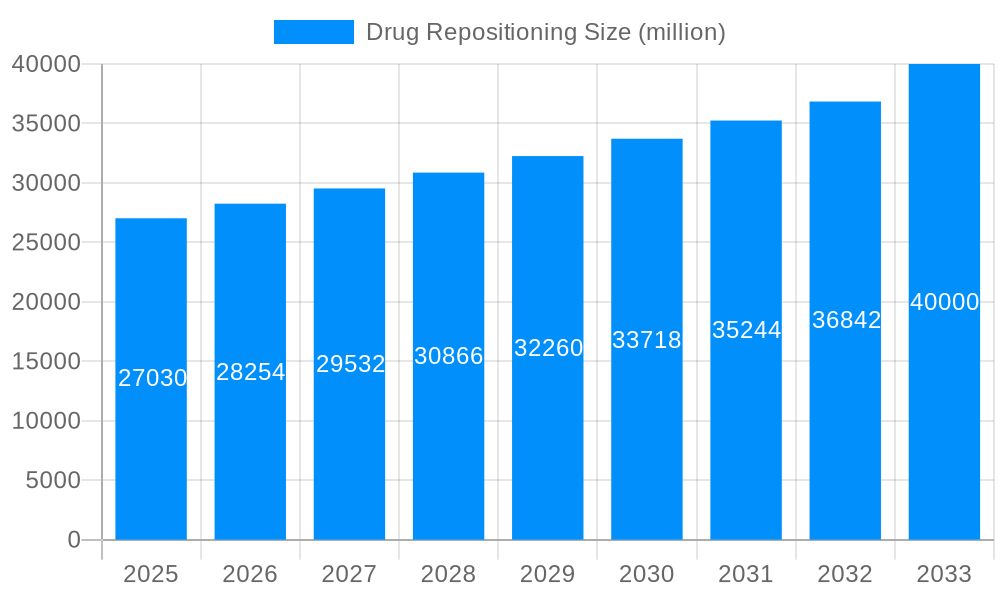

The global drug repositioning market is experiencing a period of significant transformation, driven by the increasing costs of new drug development and the unmet needs of numerous patient populations. The market, valued at USD 25 billion in 2025, is projected to witness robust growth, reaching an estimated USD 50 billion by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of approximately 8% during the forecast period (2025-2033). This growth is fueled by a convergence of factors including the escalating demand for cost-effective therapies, the rising prevalence of chronic diseases, and the accelerating adoption of advanced technologies that facilitate the identification of new therapeutic applications for existing drugs. The historical period (2019-2024) saw a steady rise in interest and investment in drug repositioning, laying the groundwork for the substantial expansion predicted for the coming decade. Key market insights reveal a growing preference for generic drugs within the repositioning landscape, driven by their lower cost and established safety profiles. Furthermore, the application of repositioned drugs in the treatment of cardiovascular diseases and cancer is currently dominating the market, reflecting the significant unmet needs within these therapeutic areas. The successful repositioning of drugs that previously failed clinical trials is also contributing to market expansion, showcasing the value of repurposing existing compounds. Companies like Teva, Novartis, and Pfizer are actively involved, leading the charge in research, development, and commercialization. This strategic shift towards drug repositioning promises to revolutionize healthcare access, accelerating the development of affordable and effective treatments for a wider range of diseases.
Several powerful forces are propelling the growth of the drug repositioning market. Firstly, the exorbitant cost of bringing a new drug to market is a major deterrent, often exceeding billions of dollars. Drug repositioning offers a significantly more cost-effective alternative, shortening the development timeline and reducing associated expenses. Secondly, the growing prevalence of chronic diseases like cancer, cardiovascular diseases, and mental illnesses is generating an urgent demand for effective and accessible therapies. Drug repositioning offers a swift pathway to meet this demand by repurposing existing drugs with established safety profiles for new therapeutic applications. Thirdly, technological advancements in bioinformatics, data analytics, and artificial intelligence are drastically improving the identification of potential drug candidates for repositioning. These tools enable researchers to sift through vast datasets and uncover previously unknown therapeutic potentials of existing drugs. Finally, regulatory incentives and streamlined approval pathways are encouraging pharmaceutical companies to invest more in drug repositioning research. A faster route to market increases the potential return on investment (ROI), making repositioning a more attractive prospect compared to traditional de novo drug discovery. These factors collectively create a robust environment for the continued growth and expansion of this promising market segment.
Despite its considerable potential, the drug repositioning market faces several challenges and restraints that could impede its growth. One major hurdle is the difficulty in securing intellectual property (IP) protection for repositioned drugs. Since the original drug's chemical structure is already known, establishing novelty and patentability can be challenging, potentially limiting the profitability of the endeavor. Furthermore, conducting thorough clinical trials to validate the efficacy and safety of a drug for a new indication can be both time-consuming and expensive, particularly in the case of niche applications. The need for extensive preclinical studies to assess potential off-target effects and interactions with other drugs also adds to the complexity and cost. Moreover, achieving regulatory approval for a new indication can be a lengthy process requiring rigorous documentation and substantial investment in clinical data generation. Finally, there can be resistance from clinicians and healthcare providers who may be hesitant to adopt a drug for a new application unless sufficient evidence of its efficacy and safety is available. Overcoming these obstacles will require a concerted effort from researchers, regulatory agencies, and healthcare providers to create a more streamlined and efficient drug repositioning process.
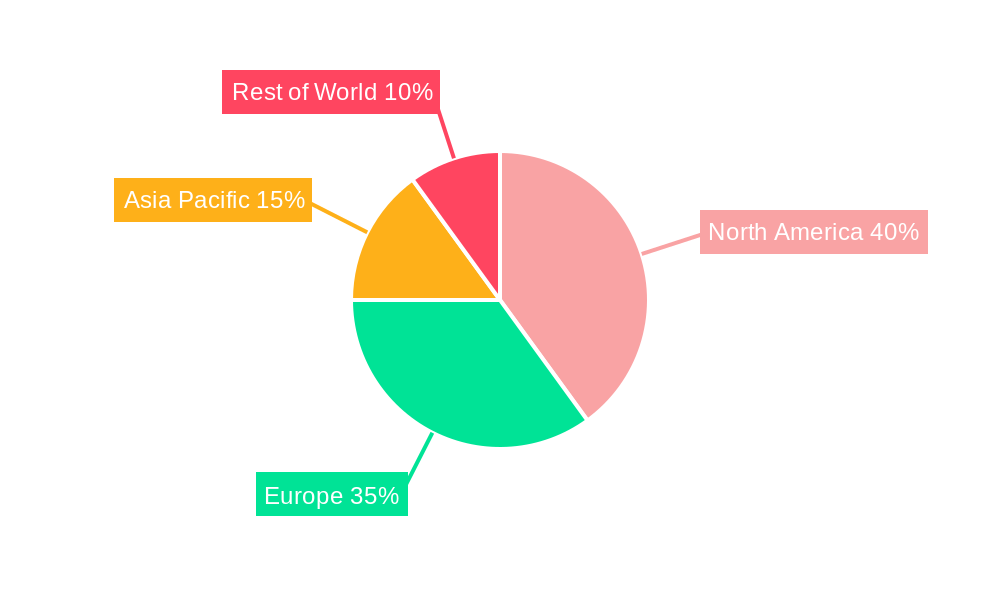
The North American market is expected to hold a dominant position in the drug repositioning landscape, driven by substantial investments in research and development, the presence of leading pharmaceutical companies, and robust regulatory frameworks. However, the Asia-Pacific region is projected to witness the fastest growth, fueled by rising healthcare expenditure, a growing prevalence of chronic diseases, and a significant unmet medical need.
Segment Domination: The segment focused on generic drugs for cardiovascular diseases is expected to dominate the market throughout the forecast period. This is largely due to the high prevalence of cardiovascular diseases globally, coupled with the cost-effectiveness of generic drugs. The established safety profiles of generic drugs and their widespread availability make them highly suitable candidates for repositioning efforts in this critical therapeutic area.
Market Dynamics within the Generic Drug Segment for Cardiovascular Diseases: The substantial patient population suffering from cardiovascular diseases creates a considerable market opportunity for repositioned generic drugs. These drugs, often offering comparable efficacy to brand-name counterparts at a fraction of the cost, address the pressing need for affordable and accessible treatments. The streamlined regulatory pathways for generic drugs further accelerate their entry into the market, fueling segment growth. Furthermore, the continuous expansion of research and development efforts in bioinformatics and data analytics facilitates the identification of promising candidates for repositioning within the existing generic drug portfolio. This synergistic interplay between market demand, economic considerations, and technological advancements ensures the dominance of this segment. The significant research investment by key players like Teva, Mylan, and Dr. Reddy's further consolidates this position.
The convergence of technological advancements, unmet medical needs, and cost pressures in the pharmaceutical industry are acting as significant growth catalysts for drug repositioning. Improved computational tools expedite the identification of potential candidates, while the escalating costs of new drug development incentivize companies to pursue this cost-effective approach. Simultaneously, the sheer volume of patients with chronic diseases fuels the demand for innovative and readily accessible treatment solutions.
This report provides a comprehensive analysis of the drug repositioning market, encompassing market size estimations, growth forecasts, and an in-depth assessment of key trends and drivers. The analysis includes a detailed review of various segments, including by drug type, therapeutic application, and geographic region. It also profiles key players in the market, evaluating their strategies and competitive landscape. The report concludes with future growth projections and identifies potential investment opportunities in the emerging drug repositioning space.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 4.5% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 4.5%.
Key companies in the market include Teva, Novartis, Mylan, Johnson & Johnson, Pfizer, Bausch Health, GSK, ChemRar Group, Glenmark, Fujifilm, Dr. Reddy, R-Pharma, .
The market segments include Type, Application.
The market size is estimated to be USD 27030 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million.
Yes, the market keyword associated with the report is "Drug Repositioning," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Drug Repositioning, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.