1. What is the projected Compound Annual Growth Rate (CAGR) of the Digitalization in the Medical Devices?
The projected CAGR is approximately 6%.
 Digitalization in the Medical Devices
Digitalization in the Medical DevicesDigitalization in the Medical Devices by Type (/> Telemedicine, Mobile Health, Mobile Medical, Health Care Analysis, Digital Medical, Other), by Application (/> Cardiovascular Devices, Orthopedic Devices, Diagnostic Imaging, MIS, IVD, Diabetes Care, Wound Management, Dental, Nephrology, Ophthalmic Devices), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global digitalization in medical devices market is poised for significant expansion, projected to reach an estimated USD 678.88 billion by 2025, driven by a robust Compound Annual Growth Rate (CAGR) of 6% over the forecast period of 2019-2033. This substantial market value underscores the accelerating integration of digital technologies into every facet of medical device development and application. Key growth drivers include the escalating demand for advanced healthcare solutions, the pervasive need for remote patient monitoring, and the increasing prevalence of chronic diseases globally, which necessitates continuous and efficient management. Furthermore, government initiatives promoting digital health adoption and the burgeoning R&D investments by leading companies are propelling innovation and market penetration. The shift towards value-based healthcare models also encourages the adoption of digital devices that can offer improved patient outcomes and cost efficiencies.
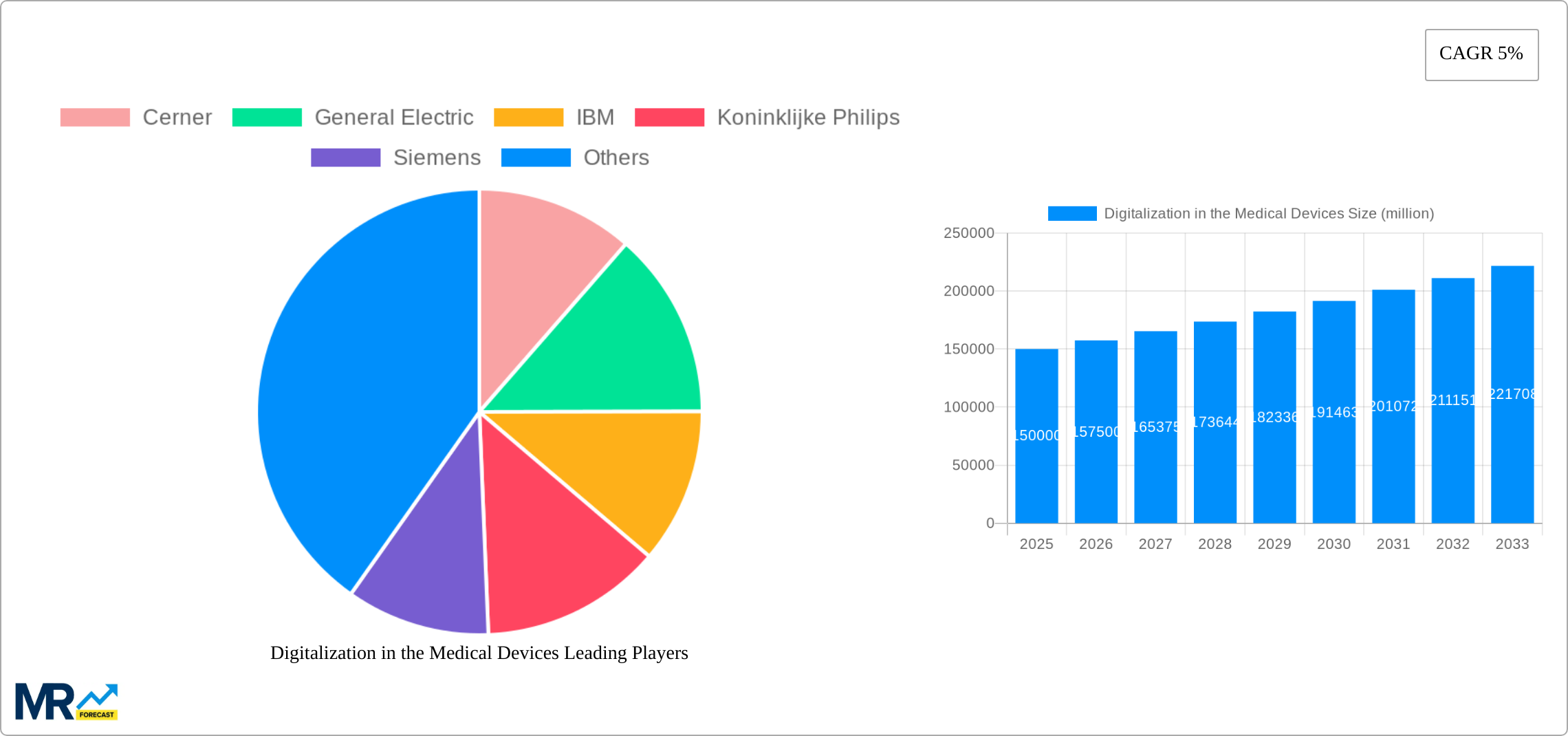

The market segmentation reveals a dynamic landscape with diverse applications and types of digital medical devices. Telemedicine and mobile health solutions are leading the charge, offering unprecedented convenience and accessibility in healthcare delivery. Applications such as cardiovascular devices, diagnostic imaging, and diabetes care are particularly benefiting from digital integration, leading to enhanced diagnostic accuracy, personalized treatment plans, and improved patient engagement. Emerging trends like the use of artificial intelligence (AI) and machine learning (ML) in medical devices for predictive analytics and personalized medicine are further shaping the market. However, challenges such as stringent regulatory frameworks, data security concerns, and the initial high cost of implementation could pose moderate restraints to the market's full potential. Despite these hurdles, the strategic investments from major technology and healthcare players, alongside a growing focus on interoperability and data standardization, are expected to pave the way for sustained growth and transformative advancements in medical device technology.
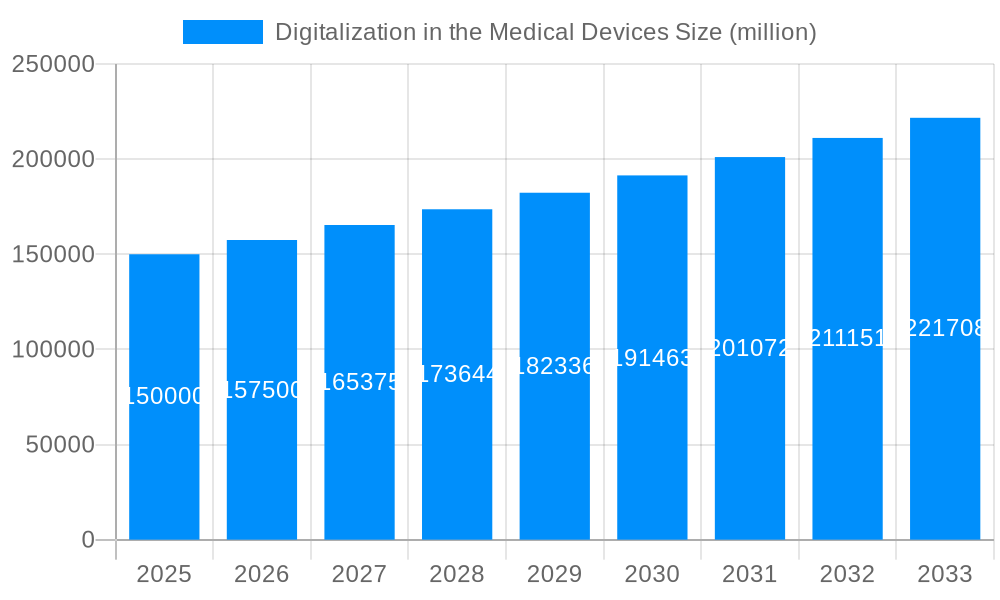

The global market for digitalization in medical devices is poised for substantial expansion, driven by an unprecedented surge in technological integration and patient-centric care models. Our analysis indicates that the market, valued at $120 billion in the base year of 2025, is projected to reach an astounding $350 billion by the end of the forecast period in 2033, exhibiting a robust Compound Annual Growth Rate (CAGR) of approximately 15% during 2025-2033. This significant growth trajectory is underpinned by a confluence of factors, including the escalating prevalence of chronic diseases, the increasing demand for remote patient monitoring solutions, and the transformative potential of artificial intelligence (AI) and machine learning (ML) in diagnostics and treatment. The historical period from 2019-2024 witnessed foundational advancements, with the market growing from an estimated $70 billion in 2019 to the base year valuation.
The current landscape is characterized by the rapid adoption of connected devices, enabling seamless data exchange between patients, healthcare providers, and medical equipment. This interconnectedness facilitates real-time health insights, proactive intervention, and personalized treatment plans. Telemedicine and mobile health applications are at the forefront of this revolution, bridging geographical barriers and democratizing access to healthcare. Innovations in wearable technology, implantable sensors, and sophisticated diagnostic imaging equipment are further augmenting the capabilities of healthcare professionals. Furthermore, the integration of cloud computing and big data analytics is empowering healthcare organizations to derive actionable intelligence from vast datasets, leading to improved operational efficiencies and enhanced patient outcomes. The emphasis is shifting from reactive care to preventative and predictive healthcare, with digital tools playing a pivotal role in identifying potential health risks before they manifest as severe conditions. The cybersecurity landscape for medical devices is also gaining prominence, with ongoing efforts to ensure the integrity and privacy of sensitive patient data. The market is witnessing a paradigm shift towards a more integrated and intelligent healthcare ecosystem.
Several powerful forces are acting as catalysts for the rapid digitalization of medical devices. Foremost among these is the escalating global burden of chronic diseases, such as diabetes, cardiovascular conditions, and respiratory illnesses. These conditions necessitate continuous monitoring and proactive management, creating a strong demand for connected devices that can provide real-time data to healthcare providers. The advent and widespread adoption of smartphones and other mobile devices have paved the way for mobile health (mHealth) solutions, allowing patients to track their health metrics, communicate with their doctors, and access health information conveniently. The growing emphasis on value-based healthcare, which prioritizes patient outcomes over the volume of services provided, is also a significant driver. Digitalization enables better tracking of outcomes, facilitates more personalized and effective treatments, and ultimately contributes to cost efficiencies for healthcare systems. Furthermore, the increasing comfort and trust in remote patient monitoring among both patients and providers, accelerated by global events like pandemics, have created fertile ground for the expansion of telehealth and remote diagnostics.
The relentless pursuit of improved patient experience and convenience is another key propellant. Patients are increasingly expecting seamless and integrated healthcare journeys, akin to their experiences in other consumer sectors. Digitalization allows for more accessible appointments, reduced waiting times, and greater patient involvement in their own care. The rapid advancements in supporting technologies, including AI, ML, IoT (Internet of Things), and cloud computing, are also critical enablers. These technologies are facilitating the development of more intelligent, predictive, and personalized medical devices. For instance, AI algorithms can analyze medical images with greater accuracy, and IoT-enabled devices can transmit vast amounts of physiological data for remote analysis. Government initiatives and supportive regulatory frameworks aimed at promoting innovation and digital health adoption are also playing a crucial role in accelerating this transformation. The growing investment by major technology companies in the healthcare sector, bringing their expertise in software development, data analytics, and user experience, is further injecting dynamism into the market.
Despite the immense potential, the digitalization of medical devices faces a complex web of challenges and restraints that can impede its widespread adoption. Foremost among these is the paramount concern for data security and patient privacy. Medical devices are increasingly connected, generating vast amounts of sensitive health information, making them prime targets for cyberattacks. Ensuring robust cybersecurity measures and compliance with stringent data protection regulations, such as GDPR and HIPAA, is a significant and ongoing challenge. The regulatory landscape for digital health solutions can be fragmented and complex, with different requirements across regions and for different types of devices. Navigating these regulatory hurdles and obtaining timely approvals can be a time-consuming and costly process for manufacturers.
Another significant restraint is the interoperability of different medical devices and health IT systems. A lack of standardization can lead to data silos, hindering the seamless exchange of information between various devices and electronic health records (EHRs). This fragmentation can limit the full potential of connected healthcare. The high cost of implementing and integrating new digital technologies can also be a barrier, particularly for smaller healthcare providers or in resource-constrained settings. While the long-term benefits are evident, the initial investment in hardware, software, and training can be substantial. Moreover, the digital literacy and comfort level of both healthcare professionals and patients can vary. Adequate training and education are crucial to ensure effective utilization of these new technologies, and resistance to change from entrenched practices can also be a factor. The ethical considerations surrounding AI-driven diagnostics and decision-making, as well as the potential for algorithmic bias, also require careful attention and mitigation strategies. Finally, ensuring the reliability and accuracy of data generated by digital medical devices is critical for clinical decision-making, and any perceived inaccuracies or malfunctions can erode trust.
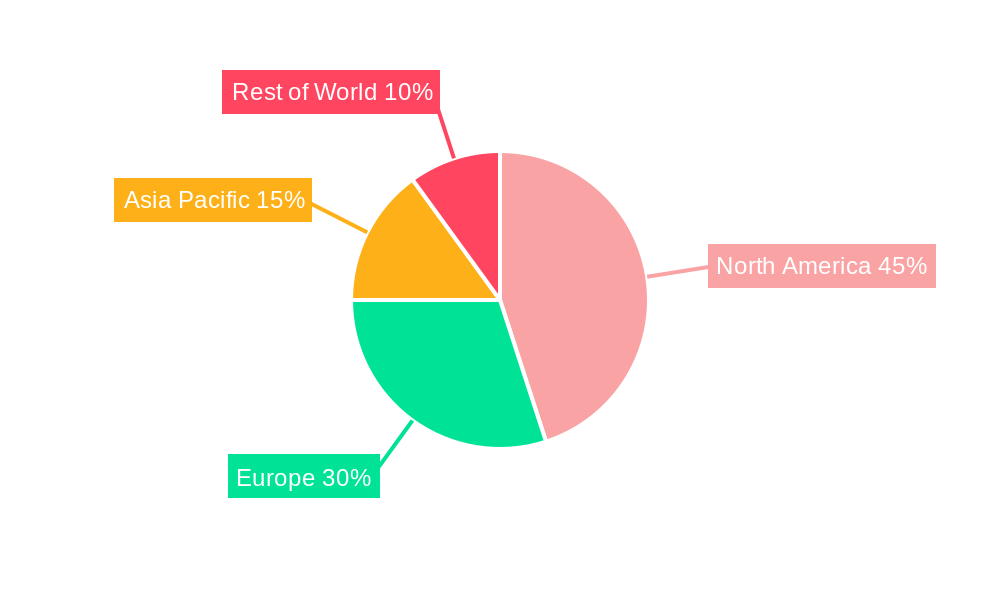
The global market for digitalization in medical devices is characterized by significant regional variations and segment dominance, driven by a complex interplay of technological adoption rates, healthcare infrastructure maturity, regulatory environments, and patient demographics.
Dominant Regions/Countries:
Dominant Segments:
The synergy between these dominant regions and segments, fueled by ongoing technological advancements and evolving healthcare needs, will continue to shape the future of digitalization in the medical devices market.
Several key growth catalysts are propelling the digitalization in the medical devices industry forward. The relentless advancements in artificial intelligence and machine learning are enabling more sophisticated diagnostics, predictive analytics, and personalized treatment plans. The widespread adoption of the Internet of Things (IoT) is facilitating seamless connectivity between devices, allowing for real-time data collection and remote monitoring, crucial for chronic disease management. Furthermore, the increasing demand for personalized medicine, driven by genomic advancements and a deeper understanding of individual patient needs, is a significant catalyst. The growing focus on preventative healthcare and early disease detection is also spurring the development of innovative digital solutions that empower individuals to take control of their well-being.
This report offers an exhaustive examination of the digitalization in the medical devices market, providing critical insights that will empower stakeholders to navigate this dynamic landscape. It delves into the intricate trends shaping the industry, highlighting the exponential growth anticipated over the study period of 2019-2033. The report meticulously analyzes the driving forces behind this digital transformation, including technological advancements and evolving healthcare demands, while also addressing the significant challenges and restraints that require strategic mitigation. Furthermore, it identifies the key regions and segments poised for dominance, offering a granular understanding of market dynamics and competitive landscapes. With comprehensive coverage of growth catalysts, leading players, and significant industry developments, this report serves as an indispensable resource for strategic decision-making, investment planning, and understanding the future trajectory of medical device digitalization.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 6%.
Key companies in the market include Cerner, General Electric, IBM, Koninklijke Philips, Siemens, Apple, AirStrip Technologies, Allscripts, Google, Orange, Qualcomm Technologies, Mqure, Samsung Electronics Co. Ltd., Telefonica S.A., Vodafone Group, McKesson Corporation.
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A.
Yes, the market keyword associated with the report is "Digitalization in the Medical Devices," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Digitalization in the Medical Devices, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.