1. What is the projected Compound Annual Growth Rate (CAGR) of the Water-based Pharmaceutical Ink?
The projected CAGR is approximately 12.08%.
 Water-based Pharmaceutical Ink
Water-based Pharmaceutical InkWater-based Pharmaceutical Ink by Type (Solids, Semi-solids, Liquids), by Application (Capsules, Interior Packaging, Exterior Packaging), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
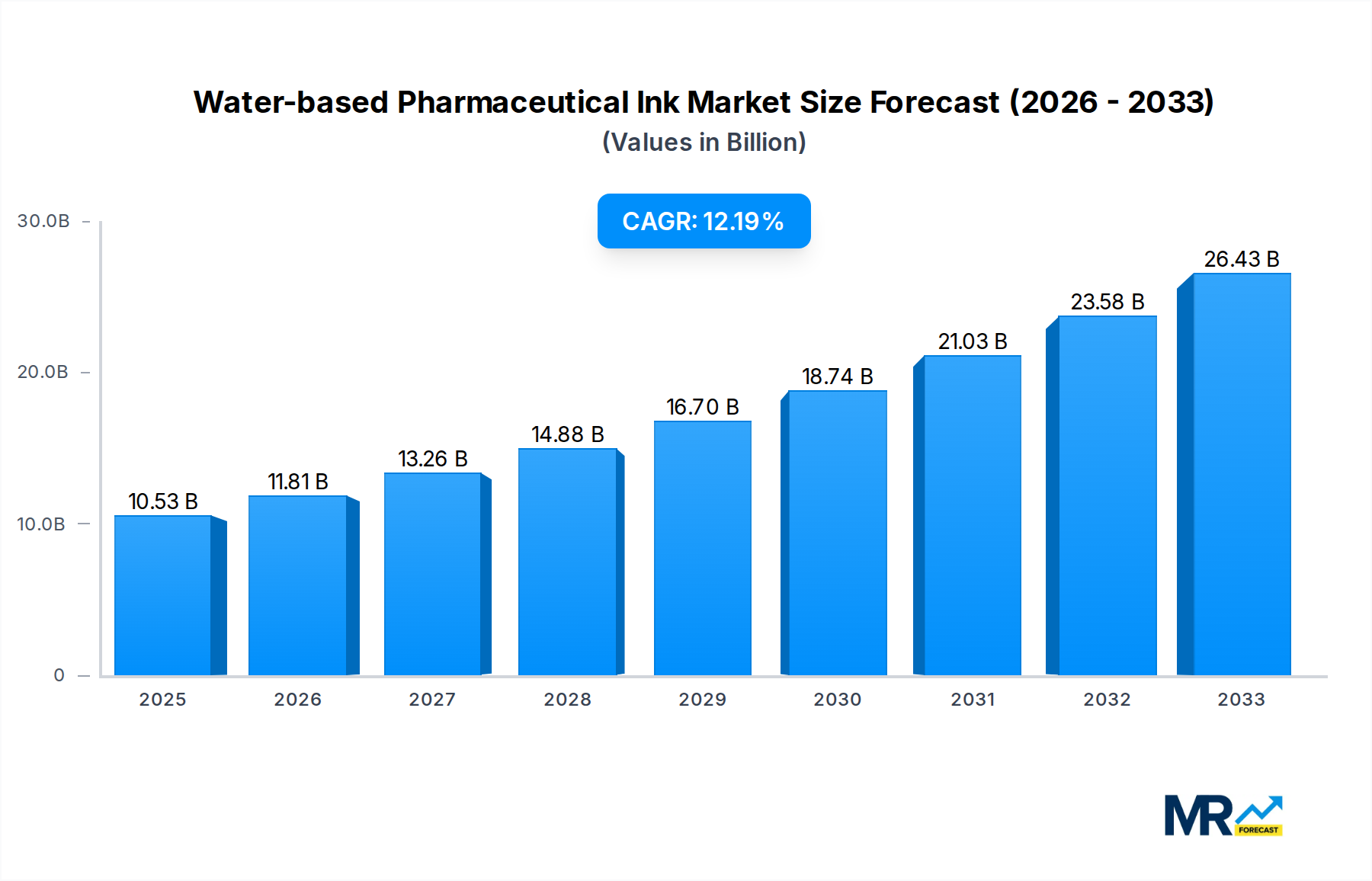
The Water-based Pharmaceutical Ink market is poised for significant expansion, projected to reach a substantial valuation of $10.53 billion by 2025, with an impressive Compound Annual Growth Rate (CAGR) of 12.08% anticipated to extend through 2033. This robust growth is primarily fueled by the increasing demand for safer and more sustainable ink solutions within the pharmaceutical industry, driven by stringent regulatory requirements and a growing emphasis on environmental responsibility. The shift away from solvent-based inks, which pose health and environmental risks, towards water-based alternatives is a pivotal factor. This transition is further bolstered by advancements in ink formulation technology, enabling enhanced printability, durability, and color vibrancy essential for pharmaceutical packaging. The market’s expansion will also be supported by the growing global pharmaceutical market itself, which necessitates a commensurate increase in packaging and labeling solutions.

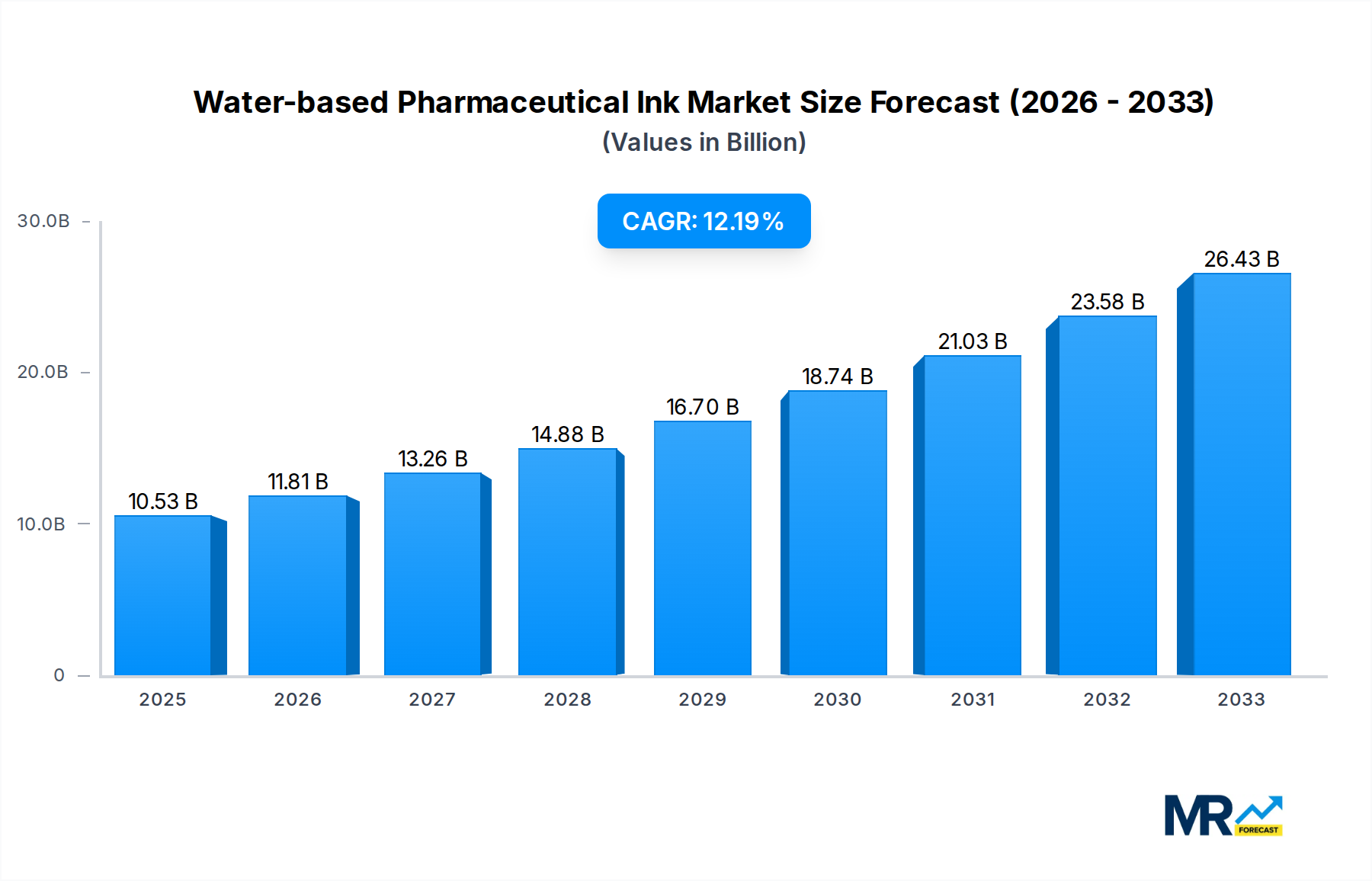
The market segments are characterized by diverse applications and types of inks. The 'Liquids' type is expected to dominate, catering to the widespread use in printing processes for various pharmaceutical packaging formats. Within applications, 'Interior Packaging' and 'Exterior Packaging' are crucial segments, requiring specialized inks for direct contact with medications or for external branding and information. Key market players such as Toyo Ink SC Holdings, Sensient Technologies, and Colorcon are actively innovating and expanding their product portfolios to meet these evolving demands. Geographically, Asia Pacific, particularly China and India, is anticipated to emerge as a significant growth engine due to its rapidly expanding pharmaceutical manufacturing base and increasing adoption of advanced packaging technologies. North America and Europe will continue to be substantial markets, driven by well-established pharmaceutical industries and a strong focus on regulatory compliance and product safety.

This comprehensive report delves into the dynamic landscape of the water-based pharmaceutical ink market, providing an in-depth analysis of its trajectory from the historical period of 2019-2024 through to the projected growth up to 2033. With the base year set at 2025, this study offers a crucial snapshot of the market's current state and forecasts its evolution. The global water-based pharmaceutical ink market is poised for substantial expansion, with the market size projected to reach $X.XX billion by 2025 and further surge to $Y.YY billion by 2033, exhibiting a compound annual growth rate (CAGR) of Z.ZZ% during the forecast period of 2025-2033. This growth is underpinned by an increasing demand for safer, more sustainable, and highly functional inks that can meet the stringent regulatory requirements of the pharmaceutical industry. Innovations in ink formulations, driven by advancements in polymer chemistry and pigment technology, are enabling the development of inks with superior adhesion, durability, and chemical resistance, crucial for pharmaceutical packaging and direct drug application. The growing awareness regarding the environmental impact of traditional solvent-based inks is also a significant factor propelling the adoption of water-based alternatives. Furthermore, the increasing complexity of pharmaceutical packaging, which often includes intricate designs, variable data printing, and anti-counterfeiting features, necessitates advanced ink solutions that water-based formulations are increasingly capable of providing. The report meticulously examines market trends across various ink types, including solids, semi-solids, and liquids, and analyzes their application across capsules, interior packaging, and exterior packaging. Industry developments, regulatory shifts, and the strategic initiatives of key market players are all thoroughly investigated to offer a holistic understanding of this vital sector.
The burgeoning water-based pharmaceutical ink market is being propelled by a confluence of powerful driving forces. Foremost among these is the escalating global demand for safer and more sustainable packaging solutions. Pharmaceutical companies are under immense pressure to reduce their environmental footprint, and the shift away from volatile organic compounds (VOCs) inherent in solvent-based inks towards water-based alternatives is a critical step in this direction. This environmental consciousness is further reinforced by increasingly stringent government regulations worldwide, which are actively promoting the use of eco-friendly materials and discouraging the use of hazardous chemicals in manufacturing processes. Moreover, the pharmaceutical industry’s unwavering commitment to patient safety necessitates inks that are non-toxic, compliant with food and drug administration (FDA) guidelines, and exhibit excellent adhesion without compromising the integrity of the drug product or its packaging. The evolving needs of the pharmaceutical sector for enhanced product differentiation and anti-counterfeiting measures also play a pivotal role. Water-based inks are proving adept at delivering vibrant colors, sharp prints, and specialized functionalities like security features, thereby safeguarding brand integrity and ensuring product authenticity.
Despite the robust growth prospects, the water-based pharmaceutical ink market is not without its challenges and restraints. A primary hurdle is the perception that water-based inks may offer inferior performance characteristics compared to their solvent-based counterparts in terms of drying speed and adhesion on certain challenging substrates commonly found in pharmaceutical packaging. While significant advancements have been made in ink formulation, overcoming these long-held perceptions and demonstrating consistent, reliable performance across diverse materials remains an ongoing effort. Furthermore, the initial investment in new printing equipment or modifications to existing infrastructure to accommodate water-based inks can be a significant barrier for some manufacturers, particularly smaller enterprises. The stringent regulatory landscape, while a driver for sustainability, also presents a challenge. Ensuring that every water-based ink formulation meets the exhaustive biocompatibility and safety standards required for pharmaceutical applications demands rigorous testing and validation, which can be time-consuming and costly. Fluctuations in raw material prices, particularly for key binders and pigments, can also impact the overall cost-effectiveness of water-based inks, potentially affecting their competitive positioning against other ink technologies.
The water-based pharmaceutical ink market is characterized by a significant dominance in North America and a particularly strong performance within the Capsules segment.
North America has emerged as a leading region due to several compelling factors that contribute to its market supremacy. The region boasts a highly developed pharmaceutical industry with a strong emphasis on innovation, research, and development. This drives a continuous demand for advanced packaging solutions that can ensure product integrity, enhance patient compliance, and meet rigorous regulatory standards. The United States, in particular, is a hub for pharmaceutical manufacturing and a significant consumer of inks for its vast array of drug products. Stringent FDA regulations concerning the safety and environmental impact of packaging materials have been instrumental in accelerating the adoption of water-based inks, which are perceived as a safer and more sustainable alternative to solvent-based inks. Furthermore, the presence of major pharmaceutical companies with substantial R&D budgets allows for greater investment in cutting-edge packaging technologies, including advanced ink formulations. A well-established supply chain, coupled with a robust network of ink manufacturers and packaging converters, further strengthens North America's market position. Government initiatives promoting green manufacturing and sustainability also provide a supportive ecosystem for the growth of water-based pharmaceutical inks in this region.
Within the segments, Capsules are demonstrating remarkable dominance. This can be attributed to the increasing prevalence of oral dosage forms in the pharmaceutical industry. Water-based inks are ideally suited for printing directly onto capsule shells, providing essential information such as drug name, dosage, batch number, and expiry date. The requirements for direct contact with the drug product necessitate inks that are not only non-toxic and biocompatible but also exhibit excellent adhesion and resistance to migration. Water-based formulations, with their low VOC content and inherent safety profiles, are increasingly favored for these critical applications. The growing trend towards personalized medicine and the need for clear, durable markings on individual capsules further amplify the demand for high-quality water-based inks. The ability of these inks to deliver sharp, legible prints that can withstand the rigors of handling and storage makes them indispensable for pharmaceutical manufacturers focusing on capsule-based drug delivery systems. The continuous development of specialized water-based inks with enhanced properties like rub resistance and moisture resistance further solidifies their dominance in this lucrative segment.
The water-based pharmaceutical ink industry is experiencing significant growth, catalyzed by several key factors. The escalating global demand for sustainable and eco-friendly packaging solutions is a primary driver, as pharmaceutical companies strive to reduce their environmental impact and comply with evolving regulations. Advances in ink formulation technology are continuously improving the performance, durability, and safety of water-based inks, making them increasingly viable for a wider range of pharmaceutical applications. The growing emphasis on product serialization and anti-counterfeiting measures necessitates inks that can deliver high-resolution printing and incorporate advanced security features. Furthermore, the expanding pharmaceutical market, particularly in emerging economies, is creating new opportunities for ink manufacturers.
This report offers a comprehensive examination of the water-based pharmaceutical ink market, providing invaluable insights for stakeholders. It meticulously analyzes market dynamics, including drivers, restraints, and opportunities, across the study period of 2019-2033, with a detailed focus on the base year 2025 and the forecast period 2025-2033. The report segments the market by ink type (solids, semi-solids, liquids) and application (capsules, interior packaging, exterior packaging), offering granular data and forecasts for each. Furthermore, it highlights key industry developments and technological innovations shaping the market's future. By profiling leading players such as Toyo Ink SC Holdings, Sensient Technologies, and Colorcon, the report provides a strategic overview of the competitive landscape. This in-depth analysis ensures that readers gain a holistic understanding of the water-based pharmaceutical ink market's current status and its promising trajectory, enabling informed strategic decision-making.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 12.08% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 12.08%.
Key companies in the market include Toyo Ink SC Holdings, Sensient Technologies, Colorcon, Fuzhou Obooc Technology, Videojet Technologies, Sun Chemical, Nazdar, Markem-Imaje, Koel Colours, .
The market segments include Type, Application.
The market size is estimated to be USD 10.53 billion as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in billion and volume, measured in K.
Yes, the market keyword associated with the report is "Water-based Pharmaceutical Ink," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Water-based Pharmaceutical Ink, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.