1. What is the projected Compound Annual Growth Rate (CAGR) of the Pharmaceutical Excipients For Injection?
The projected CAGR is approximately 5.4%.
 Pharmaceutical Excipients For Injection
Pharmaceutical Excipients For InjectionPharmaceutical Excipients For Injection by Application (Chemical Injections, Traditional Chinese Medicine Injections, Others), by Type (Solvent, Solubilizers, pH Adjuster, Antioxidants, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global market for Pharmaceutical Excipients for Injection is experiencing robust growth, projected at a Compound Annual Growth Rate (CAGR) of 5.4%, with an estimated market size of USD 1576.4 million in 2025. This expansion is fueled by several key drivers, including the increasing prevalence of chronic diseases necessitating advanced injectable drug formulations, a growing demand for biopharmaceuticals and biologics which often require specialized excipients for stability and delivery, and the continuous innovation in drug delivery systems aiming for enhanced therapeutic efficacy and patient compliance. The trend towards more sophisticated parenteral formulations, such as liposomes, nanoparticles, and long-acting injectables, is a significant factor propelling market development. Furthermore, the expanding pharmaceutical R&D pipeline and the growing number of clinical trials for injectable drugs contribute to a sustained demand for high-quality excipients. The market is segmented across various applications, including Chemical Injections and Traditional Chinese Medicine Injections, and by type, encompassing critical components like Solvents, Solubilizers, pH Adjusters, and Antioxidants. These segments cater to the diverse needs of injectable drug manufacturing, from ensuring drug solubility and stability to controlling the release profile of active pharmaceutical ingredients (APIs).


The market's trajectory is further shaped by evolving regulatory landscapes that emphasize stringent quality and safety standards for injectable excipients, pushing manufacturers to invest in advanced production processes and purification technologies. Key industry players such as Dow, Nippon Fine Chemical, Croda, and Pfanstiehl are actively involved in research and development to introduce novel excipient solutions that address emerging challenges in parenteral drug formulation, such as improved bioavailability and reduced immunogenicity. While growth is strong, certain restraints may influence the pace, including the high cost of developing and validating new excipients, and the complex supply chain management required to ensure a consistent supply of high-purity materials. Geographically, the Asia Pacific region, particularly China and India, is emerging as a significant growth hub due to its expanding pharmaceutical manufacturing capabilities and rising healthcare expenditure. North America and Europe continue to be major markets, driven by established pharmaceutical industries and a strong focus on R&D for innovative injectable therapies.
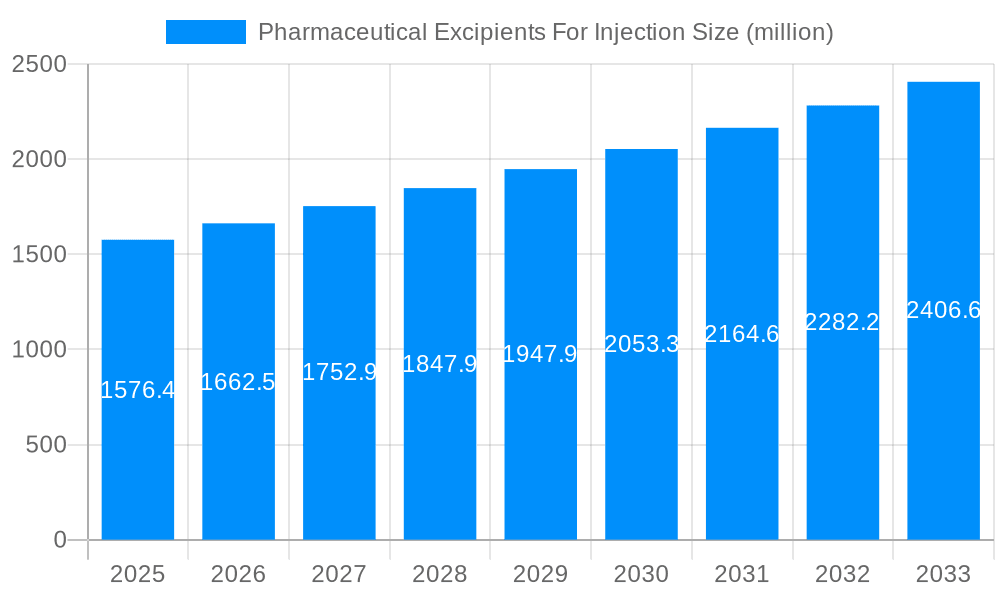

The global pharmaceutical excipients for injection market is poised for substantial expansion, driven by an escalating demand for advanced drug delivery systems and a burgeoning pharmaceutical industry, particularly in emerging economies. During the study period of 2019-2033, with a base year of 2025 and a forecast period of 2025-2033, the market is projected to witness remarkable growth. Key market insights reveal a consistent upward trajectory, influenced by a growing prevalence of chronic diseases requiring injectable treatments, a rising geriatric population, and an increasing focus on the development of novel biologic drugs. The historical period of 2019-2024 laid the groundwork for this expansion, characterized by steady innovation and increasing adoption of high-quality excipients to ensure drug stability, efficacy, and patient safety.
The market is segmented into various applications, including Chemical Injections, Traditional Chinese Medicine (TCM) Injections, and Others, each contributing uniquely to the overall market dynamics. Chemical Injections, often associated with mainstream pharmaceuticals, constitute a significant share due to the widespread use of parenteral drugs for various therapeutic areas like oncology, cardiology, and infectious diseases. TCM Injections, on the other hand, represent a rapidly growing segment, especially in Asian markets, reflecting a global resurgence of interest in traditional medicine integrated with modern pharmaceutical science. The "Others" category encompasses a diverse range of specialized applications, including diagnostic agents and vaccines, further broadening the market's scope.
The types of excipients also play a crucial role in defining market trends. Solvents, essential for dissolving active pharmaceutical ingredients (APIs) for parenteral administration, remain a cornerstone of the market. Solubilizers are gaining increasing importance with the rise of poorly soluble APIs, enabling effective formulation of potent drugs. pH adjusters are critical for maintaining drug stability and compatibility with physiological conditions, while antioxidants protect sensitive APIs from degradation. The "Others" category for excipient types includes emulsifiers, stabilizers, and preservatives, all contributing to the integrity and performance of injectable formulations. Industry developments are constantly pushing the boundaries, with a strong emphasis on sterile manufacturing processes, biocompatible excipients, and the development of advanced delivery systems like liposomes and nanoparticles, all of which are shaping the future landscape of pharmaceutical excipients for injection. The estimated market value for 2025 is projected to be in the millions, with a substantial compound annual growth rate (CAGR) anticipated over the forecast period, reflecting a robust and dynamic market.
The pharmaceutical excipients for injection market is experiencing robust growth propelled by several interconnected driving forces. Foremost among these is the ever-increasing global burden of chronic diseases such as diabetes, cardiovascular disorders, cancer, and autoimmune diseases. These conditions often necessitate long-term treatment regimens that frequently involve injectable drug formulations to ensure rapid and predictable therapeutic effects, bypassing the digestive system and delivering APIs directly into the bloodstream or target tissues. Consequently, the demand for high-quality, safe, and effective excipients that can facilitate the stable and efficient delivery of these life-saving medications is on a persistent rise.
Furthermore, the expanding elderly population worldwide represents another significant growth driver. As individuals age, their susceptibility to various health issues increases, leading to a greater reliance on injectable pharmaceuticals. This demographic shift, coupled with a growing awareness and preference for minimally invasive drug administration routes among all age groups, directly translates to a higher consumption of pharmaceutical excipients for injection. The ongoing advancements in pharmaceutical research and development, particularly in the realm of biologics and biosimilars, are also playing a pivotal role. These complex therapeutic agents often require specialized excipients to maintain their structural integrity, enhance their solubility, and ensure their safe and effective delivery, thereby fueling innovation and market expansion. The increasing investment in research and development by pharmaceutical companies, aimed at creating novel drugs and optimizing existing ones, further accentuates the need for a diverse and sophisticated range of excipients.
Despite the promising growth trajectory, the pharmaceutical excipients for injection market faces several formidable challenges and restraints that could impede its full potential. A primary concern revolves around the stringent regulatory landscape governing parenteral drug products. Regulatory bodies worldwide, such as the FDA in the US and the EMA in Europe, impose rigorous standards for the quality, purity, and safety of excipients used in injectable formulations. Obtaining regulatory approval for new excipients or for the use of existing excipients in novel applications can be a time-consuming, complex, and expensive process, often involving extensive preclinical and clinical trials. This can slow down the pace of innovation and market entry for new products.
Moreover, the inherent complexity of injectable formulations demands a high degree of purity and consistency in excipients. Any deviation from established quality standards can have severe consequences for drug efficacy and patient safety, leading to product recalls and reputational damage for manufacturers. This necessitates significant investment in sophisticated manufacturing processes, quality control measures, and robust supply chain management, which can increase operational costs. The development and validation of novel excipients with specific functionalities, such as enhanced drug solubility or controlled release properties, also require substantial research and development investments. Furthermore, the emergence of counterfeit or substandard excipients poses a significant threat to patient safety and market integrity, necessitating continuous vigilance and efforts to combat illicit trade. Geopolitical uncertainties and supply chain disruptions, as witnessed in recent global events, can also impact the availability and pricing of raw materials, posing a challenge for consistent production.
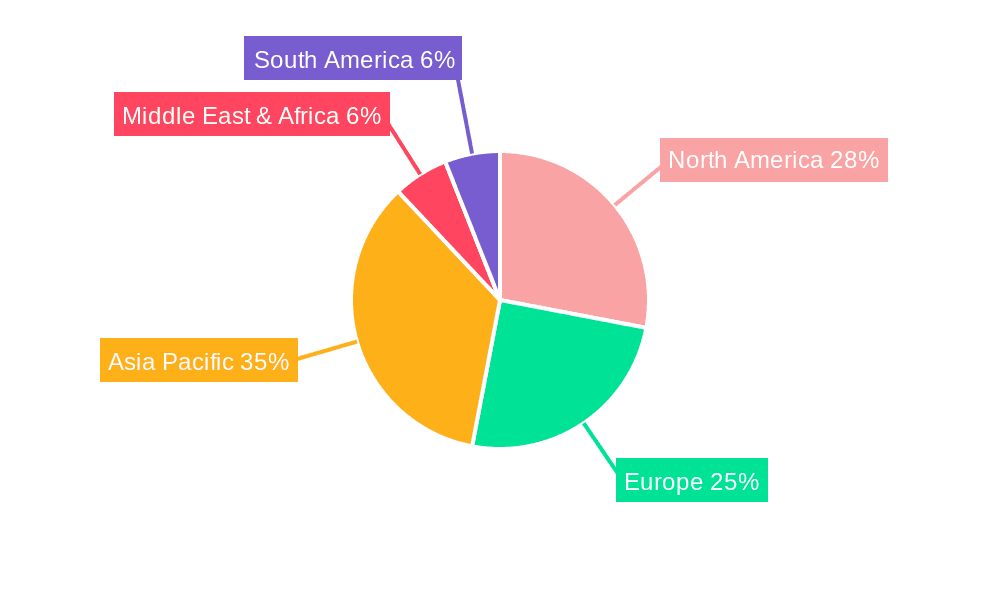
The global pharmaceutical excipients for injection market is anticipated to witness significant dominance by North America and Asia Pacific, particularly driven by the Chemical Injections application segment and the Solvent type segment.
In North America, the market's leadership is underpinned by its well-established and advanced pharmaceutical industry, characterized by substantial investments in research and development, a strong presence of major pharmaceutical and biotechnology companies, and a high prevalence of chronic diseases requiring injectable treatments. The region’s robust healthcare infrastructure, coupled with a high disposable income and a proactive approach to adopting innovative medical technologies, fuels the demand for a wide array of injectable drugs and, consequently, the excipients used in their formulation. The United States, in particular, stands as a dominant force due to its large patient pool and a consistent pipeline of novel drug approvals. The focus on developing advanced therapeutic modalities, including biologics and personalized medicine, further drives the need for specialized and high-purity excipients. The regulatory environment, while stringent, also fosters innovation by providing clear pathways for product development and approval for well-researched and safe excipients.
The Asia Pacific region, on the other hand, is projected to exhibit the most dynamic growth and is steadily gaining prominence. This surge is attributed to several factors: a rapidly expanding population, a growing middle class with increased access to healthcare, and a burgeoning pharmaceutical manufacturing sector. Countries like China and India are emerging as major hubs for both the production and consumption of pharmaceuticals, including injectable drugs. The increasing prevalence of lifestyle-related diseases and a greater emphasis on preventive healthcare are contributing to the demand for injectable medications. Furthermore, government initiatives aimed at promoting domestic pharmaceutical production and research, coupled with cost-effective manufacturing capabilities, are making the region an attractive destination for both local and global pharmaceutical companies. The rising interest and investment in Traditional Chinese Medicine (TCM) Injections within this region also contribute significantly to the overall market growth for specific types of excipients.
Focusing on the Chemical Injections application segment, its dominance is a direct reflection of the widespread use of conventional pharmaceuticals across a vast spectrum of therapeutic areas. From life-saving antibiotics and chemotherapy drugs to essential vaccines and pain management treatments, chemical injections form the backbone of modern medicine. The continuous development of new chemical entities and the genericization of existing drugs contribute to a steady and growing demand for a broad range of excipients, including solvents, pH adjusters, and stabilizers, necessary for their formulation. The sheer volume of production and consumption of these conventional injectables globally solidifies this segment's leading position.
Within the Type segmentation, Solvents are expected to remain a dominant force. Solvents are fundamental to nearly every injectable formulation, serving as the medium for dissolving active pharmaceutical ingredients (APIs) to create a stable and administrable solution. Water for injection (WFI) and various organic solvents are indispensable components in the manufacturing process. As the pharmaceutical industry continues to expand and new injectable drugs are developed, the demand for high-purity, sterile, and pharmacopoeia-compliant solvents will invariably remain elevated. The increasing complexity of APIs, some of which have poor water solubility, also drives innovation in solvent systems, including co-solvents and specialized solvent blends, to achieve effective drug delivery. This fundamental necessity for dissolution and formulation ensures the perpetual demand and dominance of solvents in the pharmaceutical excipients for injection market.
Several key growth catalysts are fueling the expansion of the pharmaceutical excipients for injection industry. The relentless pursuit of novel drug discovery, particularly in areas like biologics and targeted therapies, necessitates the development of sophisticated and specialized excipients that can ensure drug stability, efficacy, and safe delivery. The increasing global incidence of chronic diseases and the aging demographic are creating a sustained demand for injectable treatments, directly translating to higher consumption of excipients. Furthermore, advancements in drug delivery technologies, such as nanotechnology and controlled-release systems, are opening new avenues for excipient innovation and application, driving market growth.
This comprehensive report offers an in-depth analysis of the global pharmaceutical excipients for injection market, providing invaluable insights for stakeholders. It meticulously covers market size, segmentation by application (Chemical Injections, Traditional Chinese Medicine Injections, Others) and type (Solvent, Solubilizers, pH Adjuster, Antioxidants, Others), and regional dynamics. The report details industry developments, identifies key growth catalysts, and analyzes the driving forces and challenges shaping the market's trajectory. With a study period from 2019-2033, including a base year of 2025 and a forecast period from 2025-2033, the report delivers a robust outlook, supported by historical data from 2019-2024. It highlights leading market players and their contributions, offering a holistic view of this critical sector of the pharmaceutical industry.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.4% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5.4%.
Key companies in the market include Dow, Nippon Fine Chemical, Nanjing Well Pharmaceutical, Jiangsu Maxim Biological, NOF, Croda, Jiangsu Poly Pharmaceutical, Er Kang Pharmaceutical, Guangzhou Hanfang Pharmaceutical, Pfanstiehl, Xi'an Libang Pharmaceutical, Zhejiang Tianyushan Medicinal Oil, .
The market segments include Application, Type.
The market size is estimated to be USD 1576.4 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "Pharmaceutical Excipients For Injection," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Pharmaceutical Excipients For Injection, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.