1. What is the projected Compound Annual Growth Rate (CAGR) of the Medical Impurity Standards?
The projected CAGR is approximately 7%.
 Medical Impurity Standards
Medical Impurity StandardsMedical Impurity Standards by Type (Organic Standard, Inorganic Standard, World Medical Impurity Standards Production ), by Application (Food and Drink, Environment, Pharmaceutical and Life Science, Petrochemical, Others, World Medical Impurity Standards Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
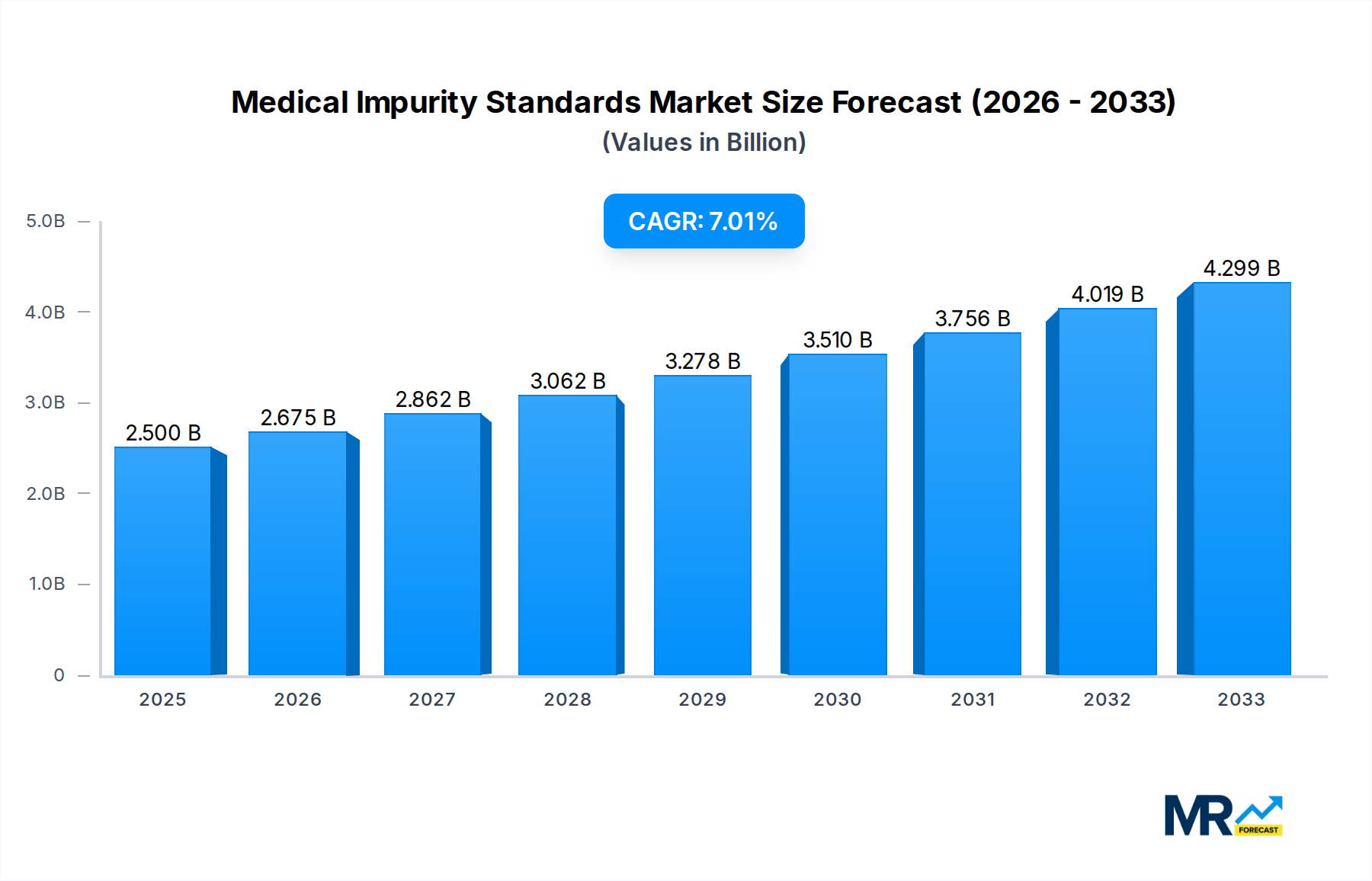
The global Medical Impurity Standards market is poised for robust expansion, projected to reach an estimated USD 2.5 billion in 2025 and grow at a Compound Annual Growth Rate (CAGR) of 7% through 2033. This significant growth is propelled by an increasing emphasis on drug safety and quality assurance across the pharmaceutical and life science sectors. Regulatory bodies worldwide are imposing stricter guidelines for the identification, quantification, and control of impurities in pharmaceutical products, thereby driving the demand for reliable and certified impurity standards. The growing complexity of drug formulations, including biologics and personalized medicines, further necessitates the availability of specialized impurity standards to ensure patient safety and therapeutic efficacy. The expansion of research and development activities, particularly in emerging economies, also contributes to the market's upward trajectory.

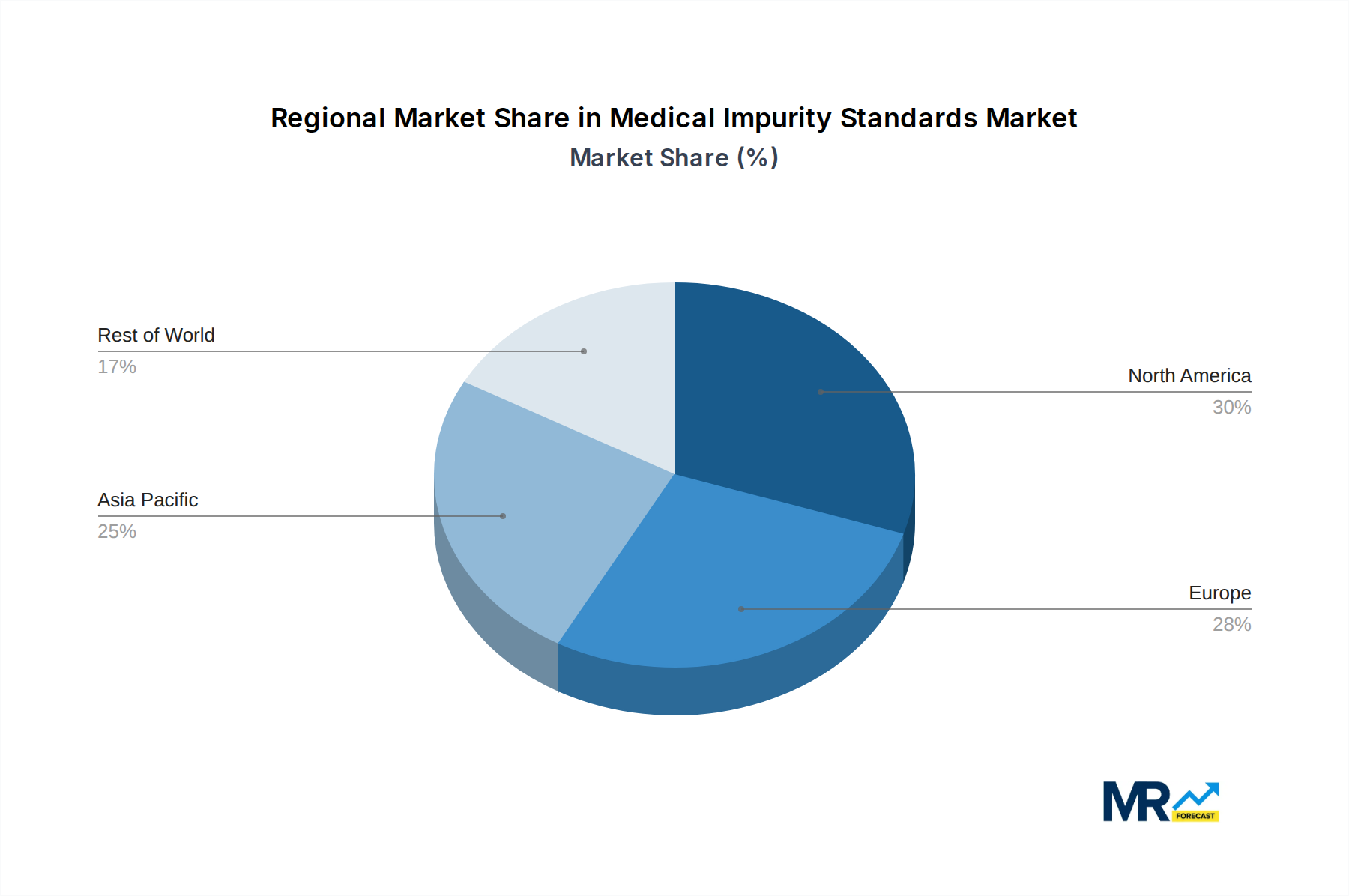
The market is segmented by type into Organic Standard and Inorganic Standard, with Organic standards likely dominating due to their relevance in characterizing a broader range of impurities found in synthetic drugs and complex biological molecules. Applications span critical sectors such as Food and Drink, Environment, Pharmaceutical and Life Science, and Petrochemical. The Pharmaceutical and Life Science segment is anticipated to hold the largest market share, driven by the stringent quality control requirements inherent in drug development and manufacturing. Leading players like Merck KGaA, LGC Standards, and Thermo Fisher Scientific are at the forefront, offering a comprehensive portfolio of high-purity impurity standards. Geographically, North America and Europe are expected to remain dominant markets, owing to established regulatory frameworks and a strong presence of pharmaceutical giants. However, the Asia Pacific region, particularly China and India, is demonstrating rapid growth, fueled by expanding pharmaceutical manufacturing capabilities and increasing investment in R&D.

Here's a comprehensive report description on Medical Impurity Standards, incorporating your specified elements and formatting:
The global Medical Impurity Standards market is undergoing a significant transformation, driven by a confluence of regulatory stringency, increasing drug development complexity, and the pervasive pursuit of patient safety. From the historical period of 2019-2024, characterized by a steady adoption of established pharmacopeial standards, we are entering an era of unprecedented demand for highly specific and sensitive impurity profiling. The base year of 2025 marks a pivotal point where the market is projected to witness substantial growth, estimated to reach new heights by the forecast period of 2025-2033. Key market insights reveal a discernible shift towards the development and utilization of standards with impurity levels quantified in the parts per billion (ppb) range, reflecting the industry's commitment to detecting even the most minute traces of potentially harmful substances. This granular approach is particularly critical in the pharmaceutical and life sciences sector, which currently dominates the application landscape. The evolving nature of synthetic chemistry, coupled with the emergence of novel therapeutic modalities like biologics and gene therapies, necessitates the continuous innovation of impurity standards to encompass a wider array of potential contaminants, including genotoxic impurities, residual solvents, and process-related impurities. Furthermore, the increasing global interconnectedness of pharmaceutical manufacturing and supply chains amplifies the need for harmonized and universally accepted impurity standards. This trend is further bolstered by regulatory bodies worldwide, who are progressively tightening their requirements for impurity identification and quantification, pushing manufacturers to invest more heavily in advanced analytical technologies and high-purity reference materials. The market's trajectory suggests a future where personalized medicine and precision therapeutics will demand even more tailored and sophisticated impurity standard solutions, moving beyond generic classifications to address compound-specific and pathway-specific impurities. The overarching insight is that the Medical Impurity Standards market is no longer just about compliance; it's a critical enabler of innovation and a fundamental pillar of global public health. The escalating complexity of pharmaceutical synthesis and the proactive approach to identifying and controlling impurities at incredibly low concentrations, often in the low to mid-hundreds of parts per billion, are setting new benchmarks. The study period from 2019 to 2033 encapsulates this dynamic evolution, with the base year of 2025 serving as a crucial indicator of accelerated growth.
The relentless pursuit of enhanced patient safety stands as the paramount driving force behind the expansion of the Medical Impurity Standards market. As regulatory agencies worldwide, including the FDA, EMA, and PMDA, increasingly scrutinize drug quality and actively enforce stringent guidelines on impurity profiling, the demand for reliable and precise impurity standards escalates. The advent of novel and complex drug molecules, particularly in the realms of biologics and advanced therapies, introduces a new spectrum of potential impurities that require dedicated reference materials for accurate identification and quantification. This complexity necessitates standards that can detect impurities at parts per billion levels, ensuring that even minute traces do not compromise therapeutic efficacy or patient well-being. Furthermore, the globalization of pharmaceutical manufacturing has amplified the need for harmonized standards across different regions, fostering greater collaboration and consistency in drug quality control. The growing awareness and concern among healthcare professionals and the general public regarding the long-term effects of impurities in pharmaceuticals also contribute to this demand. The historical period of 2019-2024 witnessed a foundational growth, but the forecast period of 2025-2033 is poised for accelerated progress, driven by these evolving dynamics.
Despite the robust growth trajectory, the Medical Impurity Standards market faces several inherent challenges that temper its full potential. The intricate nature of impurity identification and characterization often requires highly specialized expertise and sophisticated analytical instrumentation, leading to significant research and development costs for manufacturers. Developing and validating new impurity standards for emerging drug modalities, especially complex biologics and gene therapies, can be a time-consuming and resource-intensive process, often taking several years and substantial financial investment, with some niche standards costing upwards of a few hundred to a thousand parts per billion in development. Moreover, the highly regulated nature of the pharmaceutical industry necessitates stringent quality control and assurance for impurity standards themselves, adding another layer of complexity and cost to their production. Market fragmentation and the presence of a large number of smaller players can lead to price volatility and challenges in establishing universal quality benchmarks. Intellectual property rights and patent protection related to novel impurity synthesis pathways can also create barriers to entry and limit widespread adoption. Furthermore, the fluctuating global economic conditions and geopolitical uncertainties can impact investment decisions and the overall demand for these specialized chemicals. The ability to consistently source and maintain the purity of reference materials at extremely low concentrations, often in the range of tens to hundreds of parts per billion, remains a significant operational hurdle for many producers.
The Pharmaceutical and Life Science segment is poised to exert a dominant influence on the global Medical Impurity Standards market, both in terms of production and application. This dominance stems from the inherent and escalating need for robust impurity control within drug discovery, development, and manufacturing processes. Within this segment, the meticulous analysis and control of impurities are not merely a regulatory obligation but a fundamental prerequisite for ensuring the safety, efficacy, and quality of pharmaceutical products. The continuous launch of new drugs, coupled with the increasing complexity of therapeutic molecules – including small molecules, biologics, and advanced therapies like cell and gene therapies – fuels a perpetual demand for a diverse and specialized range of impurity standards. These standards are crucial for identifying and quantifying a vast spectrum of potential contaminants, such as process-related impurities, degradation products, residual solvents, and genotoxic impurities (GTIs). The stringent regulatory landscape, with bodies like the FDA and EMA mandating increasingly lower detection limits for impurities, often down to parts per billion (ppb) levels, further intensifies this demand. Companies operating in this space require standards that can accurately reflect these ultra-trace levels, pushing the boundaries of analytical chemistry and reference material production.
Furthermore, the Pharmaceutical and Life Science segment is a significant driver for innovation in the Organic Standard sub-segment. The majority of novel drug substances and their associated impurities are organic in nature, necessitating the development of highly purified organic impurity standards. This includes a wide array of complex organic molecules that require sophisticated synthesis and purification techniques to achieve the required purity levels, often in the range of hundreds to thousands of parts per billion. The geographical dominance is likely to be observed in regions with a strong pharmaceutical manufacturing base and robust research and development ecosystems. North America, particularly the United States, and Europe, with countries like Germany, Switzerland, and the United Kingdom, are expected to lead the market. This is attributed to the presence of major pharmaceutical companies, well-established regulatory frameworks, and significant investments in R&D for drug development. Asia Pacific, with its rapidly expanding pharmaceutical manufacturing capabilities and growing focus on quality compliance, is also anticipated to witness substantial market growth. The market for World Medical Impurity Standards Production itself is intrinsically linked to the Pharmaceutical and Life Science application. As the complexity and global reach of drug manufacturing increase, so does the demand for standardized, high-quality impurity reference materials produced under strict quality management systems. The production of these standards is a specialized area, often requiring expertise in analytical chemistry, organic synthesis, and stringent quality control protocols. The ability to produce standards at very low impurity concentrations, in the low to mid-hundreds of parts per billion, is a hallmark of leading manufacturers in this domain.
The Medical Impurity Standards industry is propelled by several key growth catalysts. The escalating global demand for safer and higher-quality pharmaceuticals, driven by increased patient awareness and stringent regulatory mandates worldwide, is a primary driver. The continuous innovation in drug discovery and development, particularly in the areas of biologics and personalized medicine, introduces novel impurities that necessitate the creation of new and specialized reference standards. Furthermore, advancements in analytical instrumentation, enabling the detection of impurities at extremely low concentrations, often in the parts per billion range, encourage the adoption of more refined impurity standards. The growing trend of pharmaceutical outsourcing and globalized supply chains also fuels the need for harmonized and universally recognized impurity standards.
This report offers an exhaustive exploration of the global Medical Impurity Standards market, providing in-depth analysis and insights for stakeholders. It delves into the intricate trends and dynamics shaping the industry, with a particular focus on impurity levels quantified in the parts per billion range. The study encompasses a detailed examination of the driving forces, including regulatory pressures and advancements in drug development, alongside a thorough analysis of the challenges and restraints, such as R&D costs and market fragmentation. Furthermore, the report identifies and analyzes the key regions, countries, and market segments, with a special emphasis on the dominance of the Pharmaceutical and Life Science sector and the pivotal role of Organic Standards. It also highlights significant growth catalysts and provides a comprehensive overview of leading industry players. The report's scope covers the historical period from 2019 to 2024, the base year of 2025, and projects the market's trajectory through the forecast period of 2025-2033, offering a complete understanding of the market's evolution and future potential.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 7%.
Key companies in the market include Merck KGaA, LGC Standards, Thermo Fisher Scientific, Agilent Technologies, Waters, GFS Chemicals, Spex Certiprep, Perkinelmer, Accustandard, Cayman Chemical Company, Restek, Anpel Laboratory, Horizon Discovery, Ricca Chemical Company, Altascientific, .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Medical Impurity Standards," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Medical Impurity Standards, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.