1. What is the projected Compound Annual Growth Rate (CAGR) of the Interbody Spacers?
The projected CAGR is approximately 6%.
 Interbody Spacers
Interbody SpacersInterbody Spacers by Type (Polyetheretherketone, Titanium, World Interbody Spacers Production ), by Application (Treatment of Spinal Diseases, Control Spinal Deformity Development, Protection of Spinal Nerves, Others, World Interbody Spacers Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global interbody spacers market is projected for significant expansion, driven by the escalating incidence of spinal disorders and a growing elderly demographic prone to age-related spinal degeneration. Innovations in materials science, yielding advanced biocompatible and durable spacers such as PEEK and titanium, are key growth catalysts. The increasing adoption of minimally invasive surgical techniques, which frequently employ interbody spacers for shorter recovery times and enhanced patient outcomes, is further accelerating market development. The market is segmented by material (PEEK, titanium, others), application (spinal disease treatment, deformity correction, nerve protection), and geography. While North America currently dominates due to high healthcare spending and robust medical infrastructure, the Asia-Pacific region is poised for the most rapid growth, fueled by rising healthcare awareness and increasing disposable incomes. The competitive landscape is dynamic, with leading companies like Abbott Laboratories, Zimmer Biomet, and Medtronic pursuing market share through innovation and strategic alliances. Potential restraints include the high cost of surgeries and procedural complications, which may moderate growth.
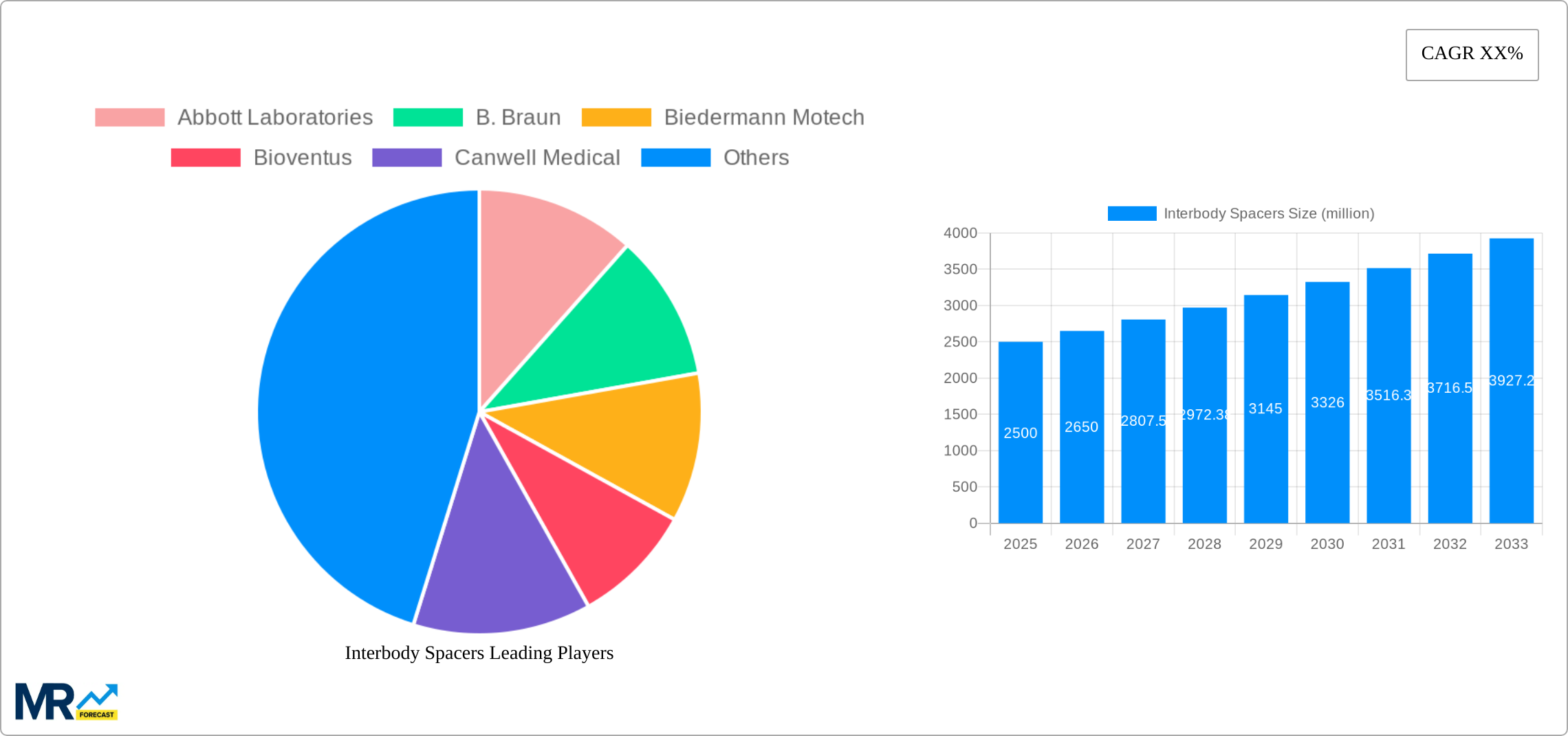

The forecast period (2025-2033) anticipates sustained growth in the interbody spacers market, primarily propelled by the increasing demand for minimally invasive spine surgeries. Market expansion will be shaped by the introduction of novel spacer designs, enhanced material properties for superior biocompatibility and longevity, and broader applications in spinal fusion procedures. Emerging markets, particularly in Asia-Pacific, represent substantial growth opportunities. However, regulatory complexities and reimbursement challenges in specific regions may present minor headwinds. Continuous research and development aimed at improving interbody spacer efficacy and safety will be instrumental in defining the market's future trajectory. A growing emphasis on patient-specific solutions and personalized medicine will also contribute to overall market advancement. The interbody spacers market size is expected to reach $2.5 billion by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of 6% from 2025.

The global interbody spacers market exhibits robust growth, projected to reach multi-million unit sales by 2033. Analysis across the study period (2019-2033), with a base year of 2025 and a forecast period spanning 2025-2033, reveals a consistently upward trajectory. This growth is fueled by several key factors, including the rising prevalence of spinal disorders globally, an aging population increasingly susceptible to age-related spinal degeneration, and advancements in minimally invasive surgical techniques. The market is witnessing a shift towards more sophisticated and biocompatible materials, particularly polyetheretherketone (PEEK) and titanium, driven by improved patient outcomes and reduced complications. Furthermore, technological innovations like 3D-printed interbody spacers are gaining traction, offering greater customization and precision. The competitive landscape is dynamic, with key players continually investing in research and development to enhance product offerings and expand market share. The historical period (2019-2024) already indicates a strong foundation for continued expansion, and the estimated year (2025) provides a benchmark for forecasting future performance. Companies are increasingly focusing on strategic partnerships and acquisitions to consolidate their position within this lucrative market. The overall trend suggests a sustained period of growth, driven by increasing demand and technological advancements within the spinal surgery sector. This expansion is not uniform across all regions or segments, with certain applications and geographical areas showing faster growth than others, suggesting opportunities for targeted market penetration. The market's resilience to economic fluctuations further underscores its long-term potential.
Several factors are significantly driving the growth of the interbody spacers market. The escalating global prevalence of spinal disorders, including degenerative disc disease, spondylolisthesis, and scoliosis, is a primary catalyst. An aging global population contributes significantly to this rise in spinal pathologies. The increasing demand for minimally invasive surgical procedures is another key driver, as interbody spacers are integral components of these techniques, offering advantages such as smaller incisions, reduced trauma, shorter hospital stays, and faster recovery times. Technological advancements in spacer design and materials science, leading to improved biocompatibility, enhanced osseointegration, and better long-term stability, are also boosting market growth. The development of advanced imaging techniques for accurate diagnosis and surgical planning further facilitates the adoption of interbody spacers. Regulatory approvals for innovative spacer designs and materials further contribute to market expansion. Finally, rising healthcare expenditure globally, especially in developed nations, provides the necessary economic environment for market growth, allowing for increased access to advanced spinal surgery.
Despite the promising growth trajectory, several challenges and restraints hinder the market's full potential. The high cost of interbody spacers, coupled with the overall expense of spinal surgery, can limit accessibility, particularly in developing countries with constrained healthcare budgets. Potential complications associated with the surgery, such as infection, nerve damage, and implant failure, can create hesitancy among patients and healthcare providers. The regulatory landscape surrounding medical devices can be complex and demanding, requiring extensive clinical trials and approvals before new products can enter the market. Stringent quality control and safety regulations add to the complexity and cost of manufacturing and distribution. Competition among established players and the emergence of new entrants can put pressure on profit margins. Finally, the lack of awareness about advanced spinal surgery techniques and the benefits of interbody spacers, especially in underserved communities, can pose a barrier to market penetration.
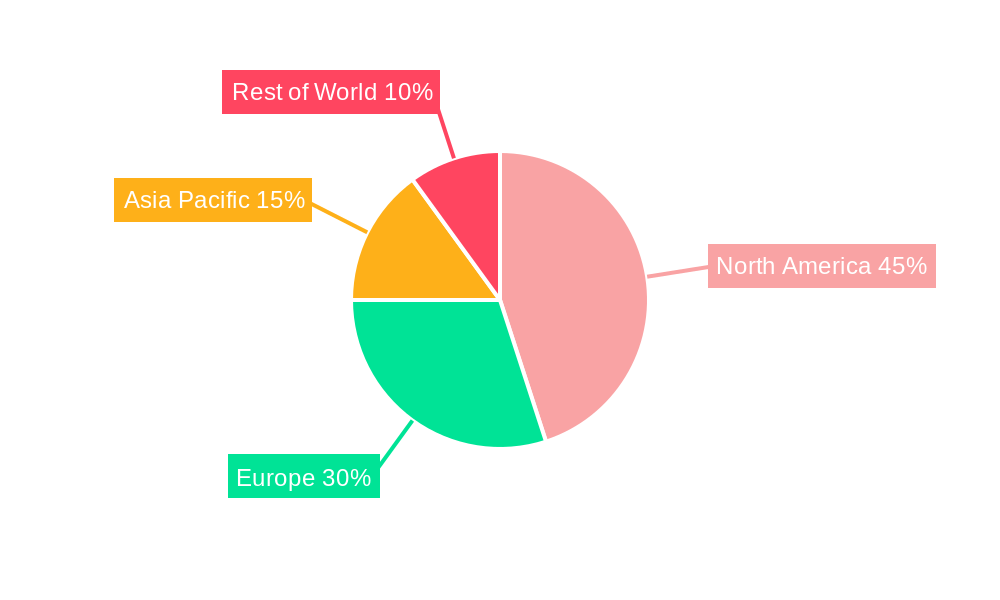
The North American market, particularly the United States, is projected to hold a significant share of the global interbody spacers market throughout the forecast period. This dominance stems from factors such as a high prevalence of spinal disorders, well-developed healthcare infrastructure, high healthcare expenditure, and early adoption of advanced surgical techniques. Europe also represents a substantial market segment, driven by similar factors although at a slightly lower pace than North America. However, the Asia-Pacific region is anticipated to witness the fastest growth rate during the forecast period, owing to a rapidly expanding elderly population, increasing healthcare spending, and rising awareness of spinal disorders.
By Type: The PEEK (Polyetheretherketone) segment is projected to dominate the market owing to its superior biocompatibility, strength, and radiolucency. Titanium spacers also hold a significant share, offering excellent mechanical properties. However, PEEK is expected to grow at a faster rate due to its superior characteristics in terms of integration with bone.
By Application: The treatment of degenerative disc disease constitutes the largest application segment due to the high prevalence of this condition. However, the application in controlling spinal deformity development and protecting spinal nerves is growing at a faster rate due to increasing awareness and advancement of minimally invasive surgeries.
The strong preference for PEEK stems from its excellent biocompatibility, reduced risk of adverse reactions, and superior osseointegration compared to other materials. Its radiolucency allows for clear visualization during post-operative imaging. The applications focusing on the prevention and correction of spinal deformities are experiencing heightened growth due to increased awareness of the long-term impact of such conditions on overall health and quality of life.
The following points summarize the key regional and segmental trends:
The interbody spacers market is propelled by a confluence of factors. Technological advancements, particularly in minimally invasive surgical techniques and material science, are leading to improved patient outcomes and increased adoption. The growing prevalence of spinal disorders, driven by an aging global population and increasingly sedentary lifestyles, fuels high demand. Increased awareness about spinal health and the effectiveness of interbody spacers is driving patient demand and encouraging physicians to utilize this technology. Finally, regulatory support and increased healthcare spending further enhance market growth.
This report provides a comprehensive overview of the interbody spacers market, analyzing historical trends, current market dynamics, and future growth prospects. It delves into key market segments, regional variations, and competitive landscapes. The report highlights significant technological advancements, regulatory changes, and emerging market opportunities, providing valuable insights for stakeholders across the industry. Detailed market sizing and forecasting are included, offering a clear picture of the market's potential.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 6%.
Key companies in the market include Abbott Laboratories, B. Braun, Biedermann Motech, Bioventus, Canwell Medical, ulrich medical, Zimmer Biomet, NuVasive, Spinal Elements, Alphatec Spine, XTANT MEDICAL, Precision Spine, Johnson and Johnson, Aurora Spine, Medtronic, JMT.
The market segments include Type, Application.
The market size is estimated to be USD 2.5 billion as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in billion and volume, measured in K.
Yes, the market keyword associated with the report is "Interbody Spacers," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Interbody Spacers, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.