1. What is the projected Compound Annual Growth Rate (CAGR) of the Glyburide API?
The projected CAGR is approximately 5.2%.
 Glyburide API
Glyburide APIGlyburide API by Application (Tablets, Capsules), by Type (Purity≥99%, Purity), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The Glyburide API market is projected for robust growth, with an estimated market size of $1.2 billion in 2025, expanding at a Compound Annual Growth Rate (CAGR) of 5.2% through 2033. This expansion is primarily fueled by the increasing prevalence of type 2 diabetes globally, a condition for which Glyburide is a widely prescribed oral antidiabetic medication. The growing demand for cost-effective diabetes management solutions, coupled with the continuous need for high-purity Glyburide APIs for pharmaceutical formulations, underpins this positive market outlook. Key applications for Glyburide API are predominantly in the manufacturing of tablets and capsules, with a strong emphasis on products with purity levels of ≥99%. This signifies a market that values quality and efficacy, essential for therapeutic applications.
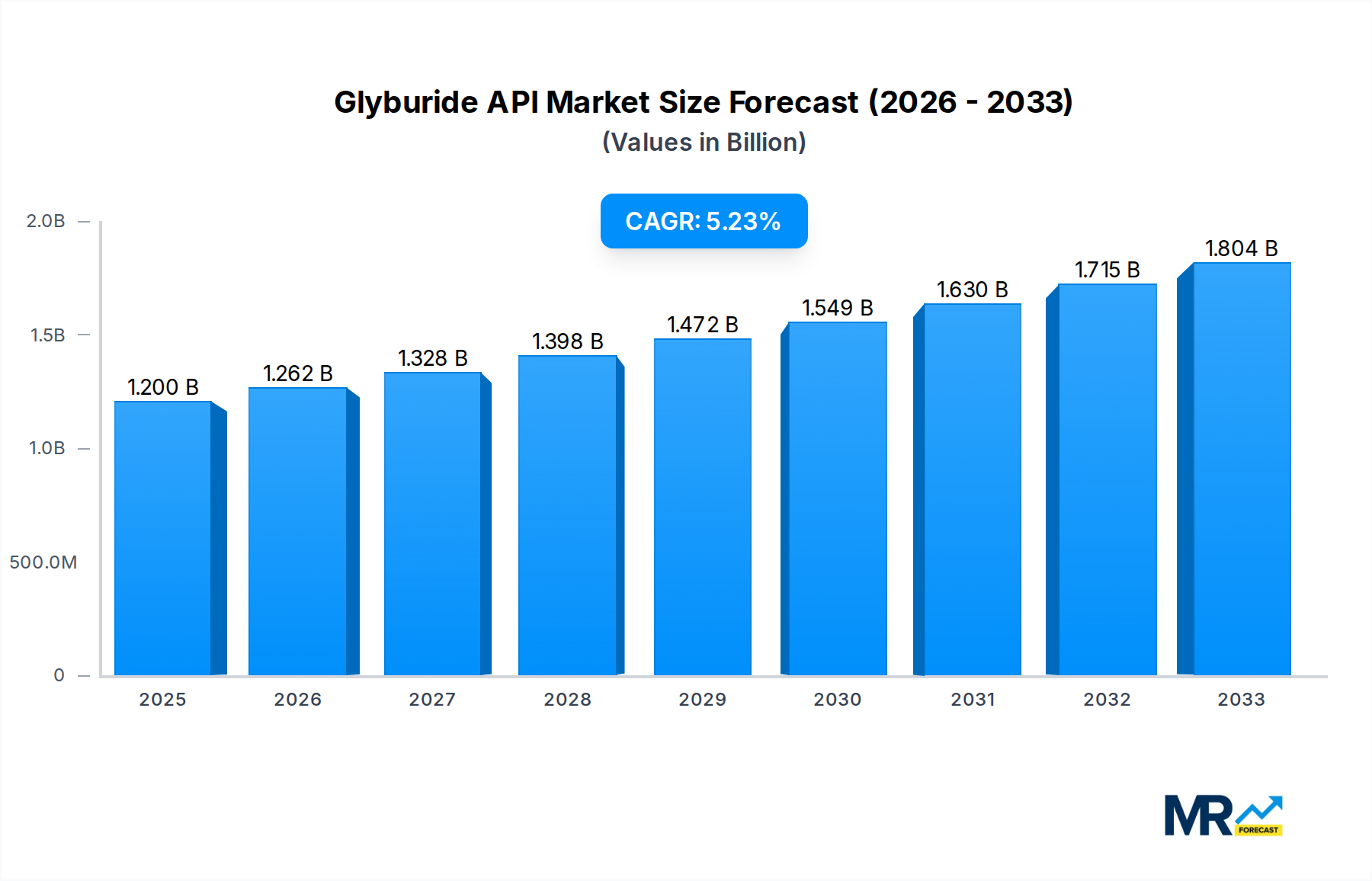

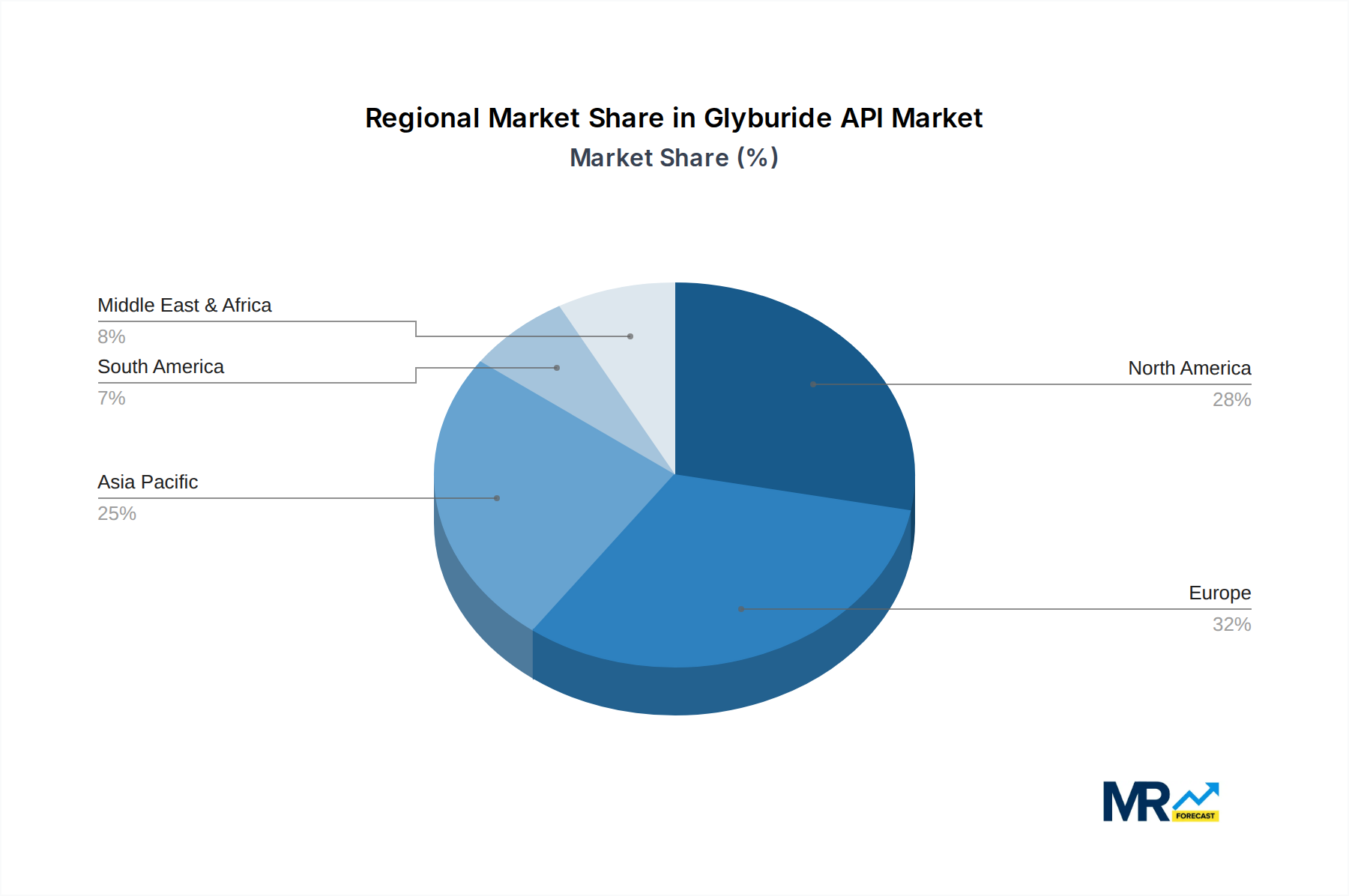
The market is characterized by a dynamic competitive landscape with established players and emerging manufacturers striving to capture market share. Strategic initiatives such as capacity expansion, product innovation, and geographical expansion are anticipated to shape the market’s trajectory. While the market is generally driven by strong demand fundamentals, potential restraints could include stringent regulatory hurdles for API manufacturing and potential shifts towards newer classes of antidiabetic drugs, though Glyburide's established efficacy and affordability are likely to maintain its significant market position. The Asia Pacific region is expected to emerge as a key growth engine due to its large diabetic population and expanding pharmaceutical manufacturing capabilities, alongside established markets in North America and Europe.

Here is a unique report description for Glyburide API, incorporating the provided details and aiming for the requested word counts and formats:
The Glyburide Active Pharmaceutical Ingredient (API) market is poised for significant evolution over the next decade, as projected by a comprehensive market analysis spanning from 2019 to 2033. The Study Period of 2019-2033, with a Base Year of 2025, and an Estimated Year also of 2025, sets the stage for understanding the trajectory of this crucial pharmaceutical component. The Forecast Period from 2025-2033 indicates a sustained interest and demand, underpinned by a robust Historical Period of 2019-2024. The market's valuation is anticipated to reach figures in the billion units, reflecting its widespread application in managing type 2 diabetes. Key market insights reveal a consistent demand for high-purity Glyburide API, with Purity ≥99% being the benchmark for pharmaceutical formulation. This stringent purity requirement is driven by the critical nature of diabetes treatment, where efficacy and safety are paramount.
The global landscape for Glyburide API manufacturing is characterized by a blend of established players and emerging contenders, each vying for market share through innovation, cost-effectiveness, and adherence to rigorous quality standards. Trends indicate a growing emphasis on backward integration by API manufacturers to ensure a stable supply chain and control over raw material costs, a factor that significantly influences overall market dynamics. Furthermore, technological advancements in synthesis processes are continuously being explored to enhance yield, reduce environmental impact, and optimize production efficiency, thereby contributing to competitive pricing. The regulatory environment, while a hurdle, also acts as a catalyst for quality improvement, pushing manufacturers towards compliance with international standards. The increasing prevalence of diabetes globally continues to be the primary driver, creating a perpetually expanding market for Glyburide API, especially in its tablet and capsule formulations. The market is also witnessing subtle shifts in demand based on regional healthcare policies and access to essential medicines, further segmenting opportunities for stakeholders. The interplay between these factors paints a picture of a dynamic and resilient market, essential for global diabetes management strategies.
The Glyburide API market is experiencing robust growth, primarily propelled by the escalating global prevalence of type 2 diabetes. This chronic condition necessitates long-term management strategies, with Glyburide remaining a cornerstone therapy for many patients due to its established efficacy and affordability. The billion-dollar valuation of this market is directly linked to the sheer volume of diabetes diagnoses worldwide, creating a consistent and substantial demand for the API. Furthermore, advancements in pharmaceutical formulation technologies are enhancing the bioavailability and therapeutic profiles of Glyburide-based medications, further stimulating its adoption. Regulatory bodies worldwide continue to approve and re-approve Glyburide formulations, reinforcing its position in treatment guidelines. The accessibility and cost-effectiveness of Glyburide API, especially compared to newer, more expensive therapeutic options, make it a vital component in healthcare systems, particularly in emerging economies where access to advanced treatments may be limited. This economic advantage ensures its continued relevance and market penetration, creating a predictable demand curve for manufacturers. The aging global population also contributes significantly, as the incidence of type 2 diabetes often increases with age, thereby expanding the patient pool requiring Glyburide-based medications.
Despite its strong market position, the Glyburide API sector faces several challenges that could potentially moderate its growth trajectory. The increasing development and adoption of newer classes of antidiabetic drugs, such as SGLT2 inhibitors and GLP-1 receptor agonists, which offer distinct mechanisms of action and potential benefits for cardiovascular and renal health, represent a significant competitive threat. These newer agents, while often more expensive, are gaining traction in clinical practice, potentially diverting market share from older drugs like Glyburide. Regulatory hurdles, including stringent quality control measures and the need for continuous compliance with evolving Good Manufacturing Practices (GMP), add to production costs and complexity for API manufacturers. The price sensitivity of the Glyburide API market, particularly in cost-conscious regions, can also act as a restraint. Manufacturers are under constant pressure to optimize production processes and maintain competitive pricing, which can limit profit margins. Moreover, the potential for adverse drug reactions and the need for careful patient monitoring can sometimes lead to prescribing caution, impacting overall demand. Finally, the patent expiry of original Glyburide formulations has led to a highly genericized market, intensifying competition among API suppliers and potentially driving down prices, thus affecting revenue growth.
The global Glyburide API market exhibits a discernible dominance within specific regions and segments, largely driven by a confluence of epidemiological factors, healthcare infrastructure, and economic considerations. The Application of Glyburide API predominantly centers around Tablets, which represent the most widely prescribed and accessible dosage form for managing type 2 diabetes. This segment's dominance is further reinforced by the inherent simplicity and cost-effectiveness of tablet manufacturing processes, making them a preferred choice for both pharmaceutical companies and healthcare providers. The Type of Glyburide API that commands the largest market share is characterized by Purity ≥99%. This stringent purity standard is non-negotiable for pharmaceutical applications, ensuring the safety and efficacy of the final drug product. Any deviation from this purity level can lead to regulatory non-compliance and pose significant risks to patient health, making manufacturers' ability to consistently achieve and verify this level of purity a critical determinant of their market standing.
Geographically, Asia Pacific is poised to emerge as a dominant region in the Glyburide API market. This is primarily attributed to the region's rapidly growing populations, coupled with a sharp increase in the incidence of type 2 diabetes. Countries like China and India, with their large patient bases and burgeoning pharmaceutical manufacturing capabilities, are key contributors to this dominance. The presence of numerous API manufacturers in these nations, including companies like DIPHARMA FRANCIS SRL, CAMBREX PROFARMACO MILANO SRL, LUSOCHIMICA SPA, USV PRIVATE LTD, CADILA PHARMACEUTICALS LTD, AUROBINDO PHARMACEUTICALS LTD, ZENTIVA KS, SRI KRISHNA PHARMACEUTICALS LTD, Orgapharm, ELIXIR PHARMA, TIANJIN PHARMACEUTICAL HOLDINGS GENCOM PHARMACY CO LTD, and Tianjin Institute of Pharmaceutical Research Co., Ltd., contributes to a competitive landscape that drives production efficiency and cost-effectiveness. These companies are strategically positioned to cater to both domestic demand and global export markets, leveraging their manufacturing prowess. Furthermore, the increasing disposable income and improving healthcare access in many Asia Pacific countries are contributing to a greater demand for diabetes management solutions, including Glyburide. The regulatory environment in these countries is also becoming more aligned with international standards, facilitating export opportunities. The lower manufacturing costs in this region also provide a competitive edge, allowing for the production of high-purity Glyburide API at competitive prices, thereby solidifying its dominance in the global market. The consistent demand for Tablets and the unwavering requirement for Purity ≥99% in this region's robust pharmaceutical industry further cement Asia Pacific's leading position.
The Glyburide API industry's growth is significantly catalyzed by the persistent and rising global burden of type 2 diabetes. This chronic disease necessitates long-term pharmacological intervention, with Glyburide remaining a widely prescribed and cost-effective treatment option. The increasing awareness and diagnosis of diabetes, particularly in emerging economies, further fuel demand for Glyburide API. Moreover, the established safety profile and extensive clinical data associated with Glyburide contribute to its continued recommendation in treatment guidelines, ensuring sustained market penetration.
This report offers an exhaustive examination of the Glyburide API market, delving into its intricate dynamics from 2019 to 2033. It provides a nuanced understanding of market trends, drivers, and challenges, projecting a valuation in the billions. The report meticulously analyzes the demand for Glyburide API in its various Applications, with a strong emphasis on Tablets and Capsules, and critically assesses the importance of Purity ≥99% as a defining Type. Key regions and countries contributing to market growth, alongside significant industry developments and leading players, are comprehensively detailed. This report serves as an indispensable resource for stakeholders seeking strategic insights and a thorough market overview.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.2% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5.2%.
Key companies in the market include DIPHARMA FRANCIS SRL, CAMBREX PROFARMACO MILANO SRL, LUSOCHIMICA SPA, USV PRIVATE LTD, CADILA PHARMACEUTICALS LTD, AUROBINDO PHARMA LTD, ZENTIVA KS, SRI KRISHNA PHARMACEUTICALS LTD, Orgapharm, ELIXIR PHARMA, TIANJIN PHARMACEUTICAL HOLDINGS GENCOM PHARMACY CO LTD, Tianjin Institute of Pharmaceutical Research Co., Ltd..
The market segments include Application, Type.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Glyburide API," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Glyburide API, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.