1. What is the projected Compound Annual Growth Rate (CAGR) of the CAR T-Cell Therapy for Multiple Myeloma?
The projected CAGR is approximately 28.6%.
 CAR T-Cell Therapy for Multiple Myeloma
CAR T-Cell Therapy for Multiple MyelomaCAR T-Cell Therapy for Multiple Myeloma by Type (Monotherapy, Combination Therapy), by Application (Multiple Myeloma, Refractory or Relapsed Multiple Myeloma), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The CAR T-cell therapy market for multiple myeloma is experiencing robust growth, projected to reach a market size of $168.7 million in 2025, expanding at a compound annual growth rate (CAGR) of 28.6% from 2025 to 2033. This significant expansion is driven by several factors. The increasing prevalence of multiple myeloma, a life-threatening blood cancer, fuels demand for effective treatment options. Furthermore, advancements in CAR T-cell technology, leading to improved efficacy and reduced side effects, are attracting significant investment and fostering wider adoption. Regulatory approvals for new CAR T-cell therapies are also contributing to market expansion. The high cost of treatment remains a significant restraint, limiting accessibility for many patients. However, ongoing research into cost-effective manufacturing processes and the potential for broader insurance coverage are mitigating this challenge. The market is segmented by treatment type (autologous vs. allogeneic), therapy stage (early vs. late-stage), and route of administration. Key players like Juno Therapeutics, Kite Pharma, Novartis, and Cellectis are leading the innovation and market penetration, driving competition and further accelerating growth. The North American market currently holds a significant share, attributed to advanced healthcare infrastructure and higher adoption rates.
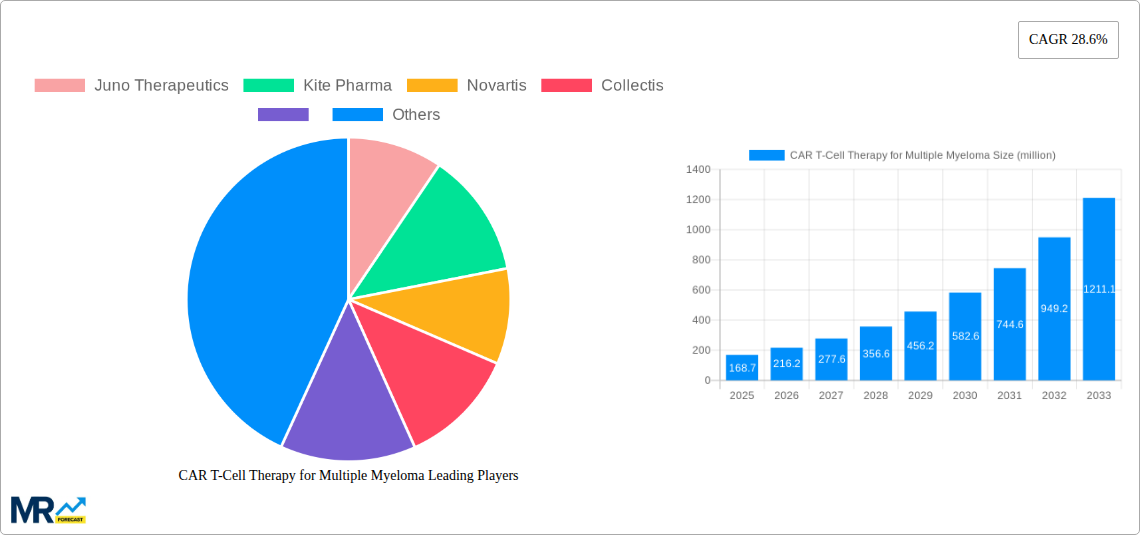

Looking forward, the market is expected to witness continued expansion driven by the development of next-generation CAR T-cell therapies. This includes research into improved targeting mechanisms, enhanced persistence, and strategies to overcome treatment resistance. The increasing focus on personalized medicine and the development of combination therapies with other treatment modalities, such as chemotherapy and immunotherapy, are poised to further revolutionize the multiple myeloma treatment landscape. Expansion into emerging markets is also anticipated, as affordability and accessibility improve, thereby broadening the reach of CAR T-cell therapy and increasing the overall market size. Despite challenges, the future outlook for the CAR T-cell therapy market in multiple myeloma remains highly positive, indicating significant growth opportunities for stakeholders throughout the forecast period.
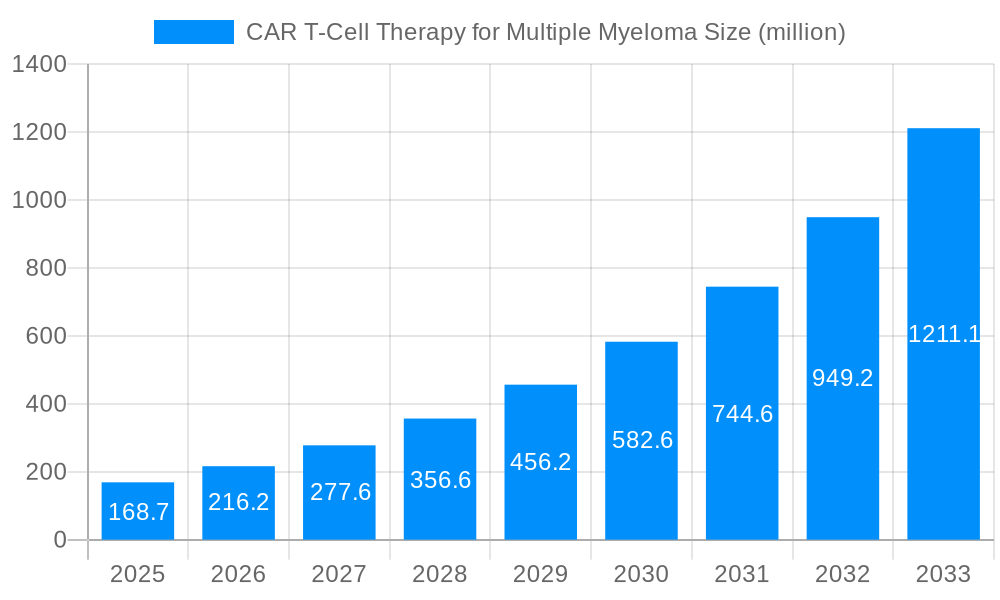

The CAR T-cell therapy market for multiple myeloma is experiencing explosive growth, projected to reach multi-billion dollar valuations by 2033. Driven by advancements in cell engineering and a greater understanding of the disease, this innovative treatment offers a lifeline to patients with relapsed or refractory multiple myeloma, a cancer of plasma cells in the bone marrow. The historical period (2019-2024) witnessed a significant increase in clinical trials and regulatory approvals, laying the groundwork for substantial market expansion. The estimated market value in 2025 is expected to be in the hundreds of millions of dollars, representing a considerable jump from the earlier years. The forecast period (2025-2033) anticipates sustained, robust growth fueled by a combination of factors including increasing patient populations, ongoing research leading to improved efficacy and safety profiles, and the expansion of reimbursement policies. While challenges remain, the overall trend strongly suggests a bright future for CAR T-cell therapy in multiple myeloma treatment. This burgeoning market is attracting significant investment from both large pharmaceutical companies and smaller biotech firms, fostering a competitive yet innovative landscape dedicated to enhancing treatment options and improving patient outcomes. The base year for our analysis is 2025, offering a critical benchmark for assessing future market trajectory. Competition is fierce, with established players and emerging companies vying for market share through technological innovation and strategic partnerships. The long-term outlook remains exceptionally positive, promising substantial improvements in the lives of patients battling this challenging malignancy.
Several key factors are propelling the growth of the CAR T-cell therapy market for multiple myeloma. Firstly, the increasing prevalence of multiple myeloma, particularly among the aging population, is creating a larger pool of potential patients. Secondly, the limitations of existing therapies, especially in relapsed/refractory settings, are driving the demand for novel and effective treatment options. CAR T-cell therapy offers a highly targeted approach, effectively eliminating myeloma cells while minimizing damage to healthy tissues. Thirdly, ongoing research and development are resulting in the development of next-generation CAR T-cell therapies with improved efficacy, safety, and durability of response. This includes exploring various CAR designs, optimizing manufacturing processes, and developing combination therapies. Furthermore, supportive regulatory environments, including accelerated approval pathways, are facilitating the quicker launch of promising new therapies. Increased investments from both private and public sectors are fueling innovation and expansion within the field. Finally, growing awareness among healthcare professionals and patients about the benefits of CAR T-cell therapy is further driving market growth. The convergence of these factors creates a powerful synergy that is propelling the adoption of this life-changing treatment modality.
Despite its immense potential, the CAR T-cell therapy market for multiple myeloma faces significant challenges. High treatment costs remain a major hurdle, making it inaccessible to many patients globally. Insurance coverage and reimbursement policies vary widely across different countries, further limiting access. The complex manufacturing process of CAR T-cell therapies adds to their expense and can lead to production bottlenecks. Moreover, side effects, such as cytokine release syndrome (CRS) and neurotoxicity, can be severe and require careful management. These adverse events can lead to hospitalization and increase the overall cost of treatment. The development of resistance to CAR T-cell therapy is also a concern, limiting the long-term effectiveness of the treatment in some patients. Finally, the need for specialized infrastructure and expertise for manufacturing, administering, and managing side effects limits the availability of this therapy to specialized centers, creating geographical disparities in access. Overcoming these challenges requires collaborative efforts across industry, regulatory bodies, and healthcare providers to improve affordability, safety, and accessibility of CAR T-cell therapy.
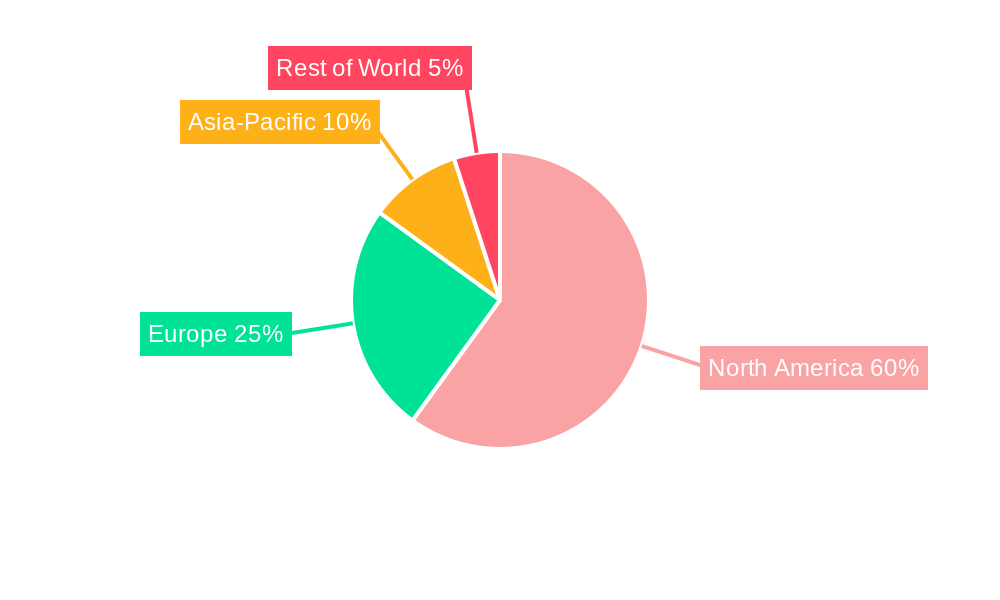
North America (US and Canada): This region is expected to dominate the market due to high healthcare expenditure, early adoption of innovative therapies, and a robust regulatory framework supporting the approval and commercialization of new drugs. The presence of major pharmaceutical companies and advanced research infrastructure further strengthens the North American market position. The US, in particular, boasts a large patient population and extensive clinical trial infrastructure, making it a pivotal market for CAR T-cell therapy development and commercialization.
Europe: European countries, especially those with well-developed healthcare systems and a focus on innovative cancer treatments, will experience significant growth. However, regulatory processes and reimbursement challenges might slightly moderate growth compared to North America.
Asia-Pacific: This region is poised for substantial growth, driven by rising healthcare expenditure, increasing awareness of advanced therapies, and expanding access to specialized healthcare facilities. However, variations in regulatory landscapes and healthcare infrastructure across different countries within this region will influence market growth rates.
Segments: The segment of relapsed or refractory multiple myeloma patients is expected to be the most significant contributor to market growth. This is due to the substantial unmet medical need in this patient population and the high efficacy of CAR T-cell therapy in extending survival and improving quality of life. Within this segment, patients exhibiting specific biomarkers that are highly responsive to CAR T-cell therapy will represent a highly targeted sub-segment with high market potential.
The overall dominance of North America reflects several factors, including robust healthcare infrastructure, well-funded research programs, and early adoption of advanced treatment modalities. The relapsed/refractory segment's dominance is driven by the urgent need for effective treatment options in this patient population, making CAR T-cell therapy an attractive choice despite its complexity and cost.
The CAR T-cell therapy market is experiencing significant growth due to several factors, including technological advancements leading to improved efficacy and safety, increased investment in research and development, expanding patient populations, and supportive regulatory environments that accelerate drug approvals. These catalysts combine to foster innovation and create a promising future for CAR T-cell therapy in treating multiple myeloma.
This report provides a detailed analysis of the CAR T-cell therapy market for multiple myeloma, encompassing market size and growth projections, key drivers and restraints, leading players, and significant industry developments. The report covers the historical period (2019-2024), base year (2025), estimated year (2025), and forecast period (2025-2033), providing a comprehensive understanding of the market’s past, present, and future trajectory. The report is an essential resource for companies operating in the industry, investors, and researchers seeking a thorough understanding of this rapidly evolving therapeutic area. The detailed segmentation analysis highlights key regional and therapeutic trends driving market growth. Furthermore, the competitive landscape assessment sheds light on the strategies employed by various companies to gain a foothold in this dynamic market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 28.6% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 28.6%.
Key companies in the market include Juno Therapeutics, Kite Pharma, Novartis, Collectis, .
The market segments include Type, Application.
The market size is estimated to be USD 168.7 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million.
Yes, the market keyword associated with the report is "CAR T-Cell Therapy for Multiple Myeloma," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the CAR T-Cell Therapy for Multiple Myeloma, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.