1. What is the projected Compound Annual Growth Rate (CAGR) of the Butyl Rubber Closure?
The projected CAGR is approximately XX%.
 Butyl Rubber Closure
Butyl Rubber ClosureButyl Rubber Closure by Type (Injection Powder Series, Frozen Dry Series, Blood Collection Series), by Application (Cartridge, Infusion Bottles, Other), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
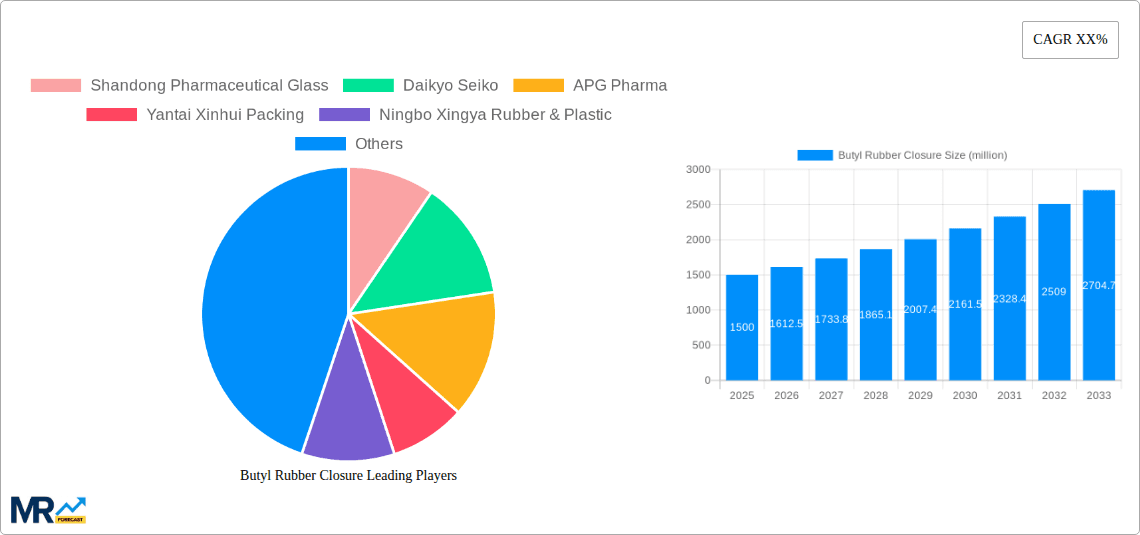
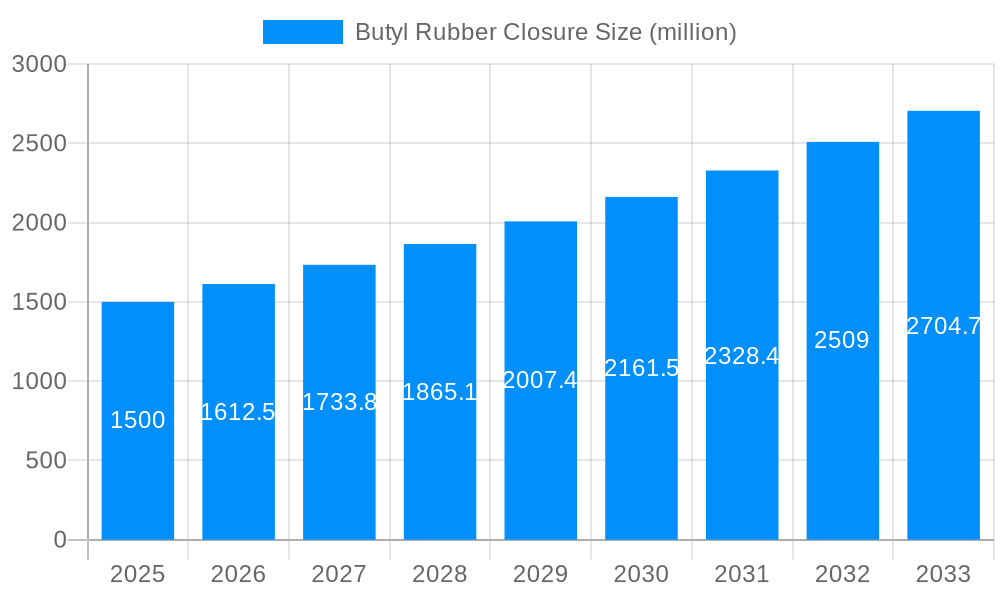
The global Butyl Rubber Closure market is poised for significant expansion, with an estimated market size of approximately $1,500 million in 2025, projecting a compound annual growth rate (CAGR) of around 7.5% through 2033. This robust growth is primarily fueled by the escalating demand for sterile and secure pharmaceutical packaging solutions, driven by the expanding global pharmaceutical industry and the increasing prevalence of chronic diseases necessitating advanced drug delivery systems. The rise in biopharmaceutical production, particularly for vaccines and biologics, further amplifies the need for high-quality butyl rubber closures that offer superior chemical resistance, low extractables, and excellent sealing properties, ensuring product integrity and patient safety. The increasing stringency of regulatory requirements regarding pharmaceutical packaging also plays a crucial role, pushing manufacturers to adopt advanced materials like butyl rubber that meet international standards.

Key market drivers include the continuous innovation in drug formulation, leading to the development of more complex and sensitive medications that require specialized packaging. The growing trend towards pre-filled syringes and vials, which rely heavily on reliable closure systems, is another significant growth catalyst. The market segments, such as Injection Powder Series and Frozen Dry Series, are expected to witness substantial demand due to their widespread application in preserving sensitive pharmaceutical ingredients. Geographically, Asia Pacific, led by China and India, is emerging as a dominant force, driven by a rapidly growing pharmaceutical manufacturing base and increasing healthcare expenditure. North America and Europe, with their mature pharmaceutical markets and high adoption rates of advanced packaging technologies, will continue to be significant revenue generators. However, challenges such as fluctuating raw material prices and the need for continuous investment in advanced manufacturing technologies could pose restraints to market growth.

Here's a unique report description on Butyl Rubber Closures, incorporating your specified details:
The global butyl rubber closure market is poised for substantial expansion, projected to reach a value exceeding 500 million units by the end of the study period in 2033. Driven by the escalating demand for sterile and secure pharmaceutical packaging solutions, the market has witnessed consistent growth across the historical period (2019-2024) and is expected to maintain this upward trajectory through the forecast period (2025-2033), with an estimated 350 million units in the base year of 2025. A significant trend shaping the market is the increasing adoption of advanced butyl rubber formulations, offering enhanced chemical resistance, lower extractables, and improved functionality for a wider range of pharmaceutical products. The rise in biologics and complex drug formulations, which often require more stringent packaging integrity, is directly fueling this demand. Furthermore, regulatory mandates emphasizing product safety and tamper-evidence are pushing manufacturers towards high-quality butyl rubber closures that meet stringent international standards. The market is also experiencing innovation in closure design, focusing on ease of use for healthcare professionals and patient safety, including features like advanced stoppers and seals that minimize contamination risks. The geographical distribution of demand is also evolving, with a growing emphasis on emerging markets in Asia-Pacific and Latin America, alongside the established markets in North America and Europe. The intricate balance between technological advancements, stringent quality controls, and the ever-increasing global healthcare needs will continue to define the landscape of the butyl rubber closure market in the coming years. The focus on sustainability in packaging is also becoming a more prominent factor, encouraging the development of more environmentally friendly butyl rubber alternatives.
The butyl rubber closure market is experiencing robust growth, primarily propelled by the insatiable global demand for safe and reliable pharmaceutical packaging. The burgeoning pharmaceutical industry, fueled by an aging global population, rising chronic disease prevalence, and significant investments in drug research and development, necessitates a constant supply of high-quality closure solutions. Specifically, the escalating production of injectable drugs, including vaccines, biologics, and biosimilars, directly translates into a heightened requirement for advanced butyl rubber stoppers and seals that ensure product sterility and prevent leakage. The increasing complexity and sensitivity of new drug formulations, such as monoclonal antibodies and gene therapies, further accentuate the need for inert and chemically stable packaging components like butyl rubber, which exhibit excellent compatibility with a wide array of active pharmaceutical ingredients (APIs). Additionally, the stringent regulatory landscape across major markets, advocating for enhanced drug safety and tamper-evidence, compels manufacturers to adopt premium butyl rubber closures that meet global pharmacopoeia standards, thereby mitigating risks of counterfeiting and product degradation. This confluence of factors creates a powerful impetus for the sustained expansion of the butyl rubber closure market.
Despite the promising growth trajectory, the butyl rubber closure market is not without its hurdles. A significant challenge lies in the stringent and evolving regulatory landscape governing pharmaceutical packaging. Manufacturers must constantly adapt to new guidelines concerning extractables and leachables, biocompatibility, and sterilization validation, which often necessitates substantial investment in research, development, and quality control. Furthermore, the volatility in raw material prices, particularly for synthetic rubber and associated chemicals, can impact the cost-effectiveness of butyl rubber closures, potentially squeezing profit margins for manufacturers and leading to price fluctuations for end-users. The increasing competition from alternative closure materials, although currently less prevalent for high-barrier applications, poses a latent threat. While butyl rubber offers superior performance in many critical applications, ongoing research into advanced polymer materials could eventually present viable substitutes. Another critical restraint is the potential for supply chain disruptions, which can arise from geopolitical instability, natural disasters, or global health crises, impacting the availability of raw materials and finished products. Finally, technical challenges in achieving absolute batch-to-batch consistency in complex butyl rubber formulations, especially for highly sensitive pharmaceutical applications, require sophisticated manufacturing processes and rigorous quality assurance protocols.
The Frozen Dry Series segment is anticipated to exert significant dominance within the global butyl rubber closure market, projected to account for a substantial portion of the market share, estimated at over 150 million units in the forecast period. This segment's leadership is intrinsically linked to the burgeoning demand for lyophilized (freeze-dried) drugs, a critical formulation method for highly sensitive biologics, vaccines, and complex therapeutic agents that require enhanced shelf-life and stability. The inherent properties of butyl rubber, including its excellent impermeability to moisture and gases, low gas transmission rate, and chemical inertness, make it an ideal choice for lyophilized products. These closures prevent the ingress of moisture and oxygen, which can degrade the active pharmaceutical ingredients during storage and transit, thereby preserving the efficacy and potency of the drug. The global increase in the production and consumption of these advanced biopharmaceuticals directly translates into a higher demand for specialized butyl rubber closures that can withstand the demanding conditions of lyophilization cycles and maintain a sterile seal post-lyophilization.
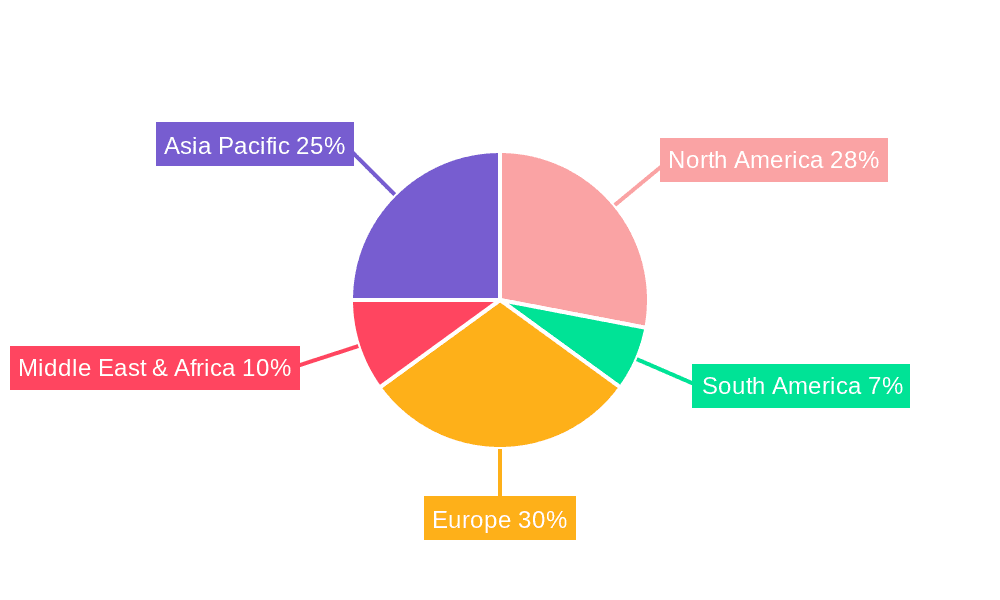
Furthermore, North America is projected to remain a dominant region in the butyl rubber closure market, driven by its well-established pharmaceutical industry, high healthcare expenditure, and a strong focus on innovation and R&D. The region's robust demand for sterile injectable drugs, coupled with stringent regulatory requirements that favor high-quality packaging solutions, positions it as a key consumer of butyl rubber closures. The presence of leading pharmaceutical and biotechnology companies, investing heavily in the development of novel therapeutics, further bolsters this dominance.
The Injection Powder Series segment is also expected to witness substantial growth, driven by the increasing use of dry powder formulations for various therapeutic areas, offering advantages in terms of stability and ease of administration. The need for secure sealing to prevent moisture contamination is paramount for these products, making butyl rubber closures a preferred choice.
In terms of Applications, Infusion Bottles are a significant driver for butyl rubber closures. The extensive use of intravenous therapies across various medical settings globally ensures a consistent demand for reliable stoppers and seals that maintain the sterility and integrity of infusion solutions.
The market is characterized by the presence of established players and emerging manufacturers, all striving to meet the evolving needs of the pharmaceutical industry. Key companies such as West Pharmaceutical, Daikyo Seiko, and UD Pharma Rubber Products are at the forefront of innovation, offering a diverse range of butyl rubber closure solutions tailored to specific drug formulations and packaging requirements. Their continuous efforts in product development, quality enhancement, and capacity expansion are crucial in meeting the escalating global demand.
The butyl rubber closure industry is significantly propelled by the escalating global demand for advanced pharmaceutical packaging solutions, particularly for biopharmaceuticals and vaccines. The increasing complexity of drug formulations, requiring inert and highly impermeable packaging, directly favors butyl rubber's superior properties. Furthermore, stringent regulatory mandates emphasizing product integrity and patient safety act as powerful catalysts, driving the adoption of high-quality butyl rubber closures that meet international standards. The burgeoning healthcare infrastructure in emerging economies also presents substantial growth opportunities.
This comprehensive report delves into the intricate dynamics of the global butyl rubber closure market, providing an in-depth analysis across its entire value chain. It meticulously examines market trends, identifies key drivers and restraints, and forecasts future market trajectories. The report offers detailed segmentation by type (Injection Powder Series, Frozen Dry Series, Blood Collection Series), application (Cartridge, Infusion Bottles, Other), and geographical region. It further scrutinizes industry developments, growth catalysts, and the competitive landscape, featuring a detailed profiling of leading market players. With a study period spanning from 2019 to 2033, and focusing on a base year of 2025, this report offers invaluable insights for stakeholders seeking to understand and capitalize on the evolving opportunities within this critical sector of pharmaceutical packaging.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XX% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately XX%.
Key companies in the market include Shandong Pharmaceutical Glass, Daikyo Seiko, APG Pharma, Yantai Xinhui Packing, Ningbo Xingya Rubber & Plastic, West Pharmaceutical, UD Pharma Rubber Products, Sagar Rrubber, GCL Pharma, .
The market segments include Type, Application.
The market size is estimated to be USD XXX million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "Butyl Rubber Closure," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Butyl Rubber Closure, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.