1. What is the projected Compound Annual Growth Rate (CAGR) of the 5-Fluocytosine API?
The projected CAGR is approximately 4.3%.
 5-Fluocytosine API
5-Fluocytosine API5-Fluocytosine API by Application (5-Fluocytosine Tablet, 5-Fluocytosine Capsule, 5-Fluocytosine Injection), by Type (0.99, 0.985, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global 5-Fluocytosine API market is poised for steady growth, exhibiting a Compound Annual Growth Rate (CAGR) of 4.3% and projected to reach an estimated market size of approximately USD 311.5 million by 2033. This growth is underpinned by increasing incidences of serious fungal infections, particularly in immunocompromised patient populations such as those undergoing chemotherapy, organ transplantation, or living with HIV/AIDS. The rising global prevalence of invasive fungal diseases, coupled with advancements in diagnostic capabilities, is driving demand for effective antifungal agents like 5-Fluocytosine. Furthermore, the expanding healthcare infrastructure in emerging economies and increased awareness regarding the management of opportunistic infections are contributing significantly to market expansion. The pharmaceutical industry's continuous investment in research and development for novel drug delivery systems and formulations of 5-Fluocytosine is also expected to fuel market progression.
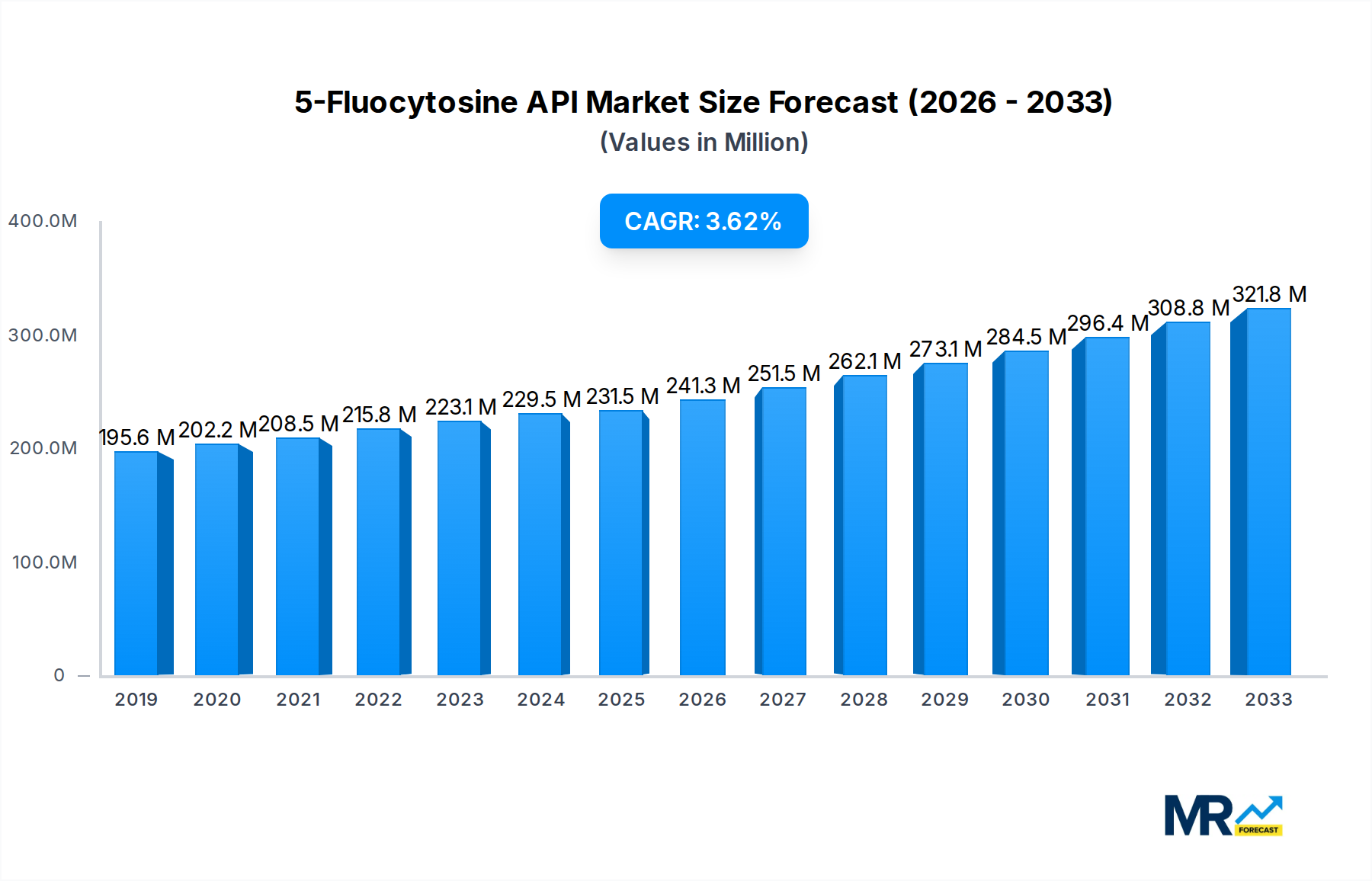

The market segmentation reveals distinct growth trajectories for various applications and purity types. The 5-Fluocytosine Injection segment is anticipated to witness robust demand owing to its rapid onset of action and suitability for severe systemic infections. While specific market share data for purity types like 0.99 and 0.985 is not explicitly detailed, higher purity grades are generally preferred in pharmaceutical applications due to stringent regulatory requirements and patient safety considerations, suggesting a potential lean towards these segments. Key players such as Jolly Healthcare, Manus Aktteva Biopharma LLP, and VIVAN Life Sciences are actively involved in the manufacturing and supply of 5-Fluocytosine API, fostering a competitive landscape. Strategic collaborations, capacity expansions, and a focus on quality compliance will be crucial for these companies to capitalize on the evolving market dynamics and address the growing global need for effective antifungal therapies.

This comprehensive report delves into the dynamic landscape of the 5-Fluocytosine API (Active Pharmaceutical Ingredient) market, offering in-depth analysis and actionable insights for stakeholders. Spanning a significant study period from 2019 to 2033, with a foundational Base Year of 2025 and an Estimated Year also of 2025, the report meticulously examines historical trends during 2019-2024 and projects future growth trajectories throughout the Forecast Period of 2025-2033. The market's valuation is presented in million units, reflecting its substantial economic impact.
The 5-Fluocytosine API market is currently experiencing a nuanced evolutionary phase, characterized by a steady demand driven by its critical role in antifungal therapies. During the Historical Period (2019-2024), the market demonstrated consistent growth, largely fueled by the persistent prevalence of systemic fungal infections and the increasing awareness surrounding their management. The API's efficacy against a spectrum of fungi, particularly Candida and Cryptococcus species, has solidified its position as a cornerstone in treating serious mycoses, often in combination with other antifungal agents. The Base Year (2025) is anticipated to reflect this sustained demand, with market players focusing on optimizing production efficiencies and ensuring a reliable supply chain.
Looking ahead into the Forecast Period (2025-2033), the market is projected to witness moderate yet significant expansion. Key insights indicate a growing trend towards the development of combination therapies, where 5-Fluocytosine API plays a synergistic role, thereby expanding its application potential and consequently its market value. Furthermore, advancements in pharmaceutical manufacturing technologies are expected to enhance the purity and consistency of the API, potentially leading to higher-value product offerings and improved therapeutic outcomes. The increasing healthcare expenditure in emerging economies, coupled with a rising incidence of immunocompromised individuals (due to conditions like HIV/AIDS, organ transplantation, and chemotherapy), will continue to be a fundamental driver. The market is also observing a subtle shift towards higher purity grades, such as 0.99 and 0.985, as regulatory stringentness and therapeutic requirements evolve. The emergence of novel delivery systems and formulations could further invigorate market dynamics. The strategic positioning of key manufacturers, including Jolly Healthcare, Manus Aktteva Biopharma LLP, and VIVAN Life Sciences, in catering to these evolving demands will be crucial for market leadership. The overall market sentiment suggests a stable and progressively growing environment for 5-Fluocytosine API, underscoring its enduring importance in addressing significant global health challenges related to fungal infections.
The sustained and projected growth of the 5-Fluocytosine API market is propelled by a confluence of powerful drivers, primarily rooted in the escalating global burden of fungal infections. The increasing prevalence of immunocompromised patient populations, stemming from factors such as the rising incidence of HIV/AIDS, a growing number of organ transplant recipients, and the widespread use of immunosuppressive therapies in cancer treatment, directly translates to a higher demand for effective antifungal agents like 5-Fluocytosine API. These infections are often severe and life-threatening, necessitating potent and reliable therapeutic options. Furthermore, the inherent efficacy of 5-Fluocytosine API as a broad-spectrum antifungal, particularly its synergistic action when combined with other antifungals like Amphotericin B, makes it an indispensable component in treating serious systemic mycoses that are difficult to manage with monotherapy. This dual action – addressing severe infections and enhancing the effectiveness of other treatments – provides a robust foundation for its market demand. The ongoing research and development efforts, though perhaps not leading to entirely new blockbuster drugs, are continuously refining the applications and understanding of existing antifungal agents, including 5-Fluocytosine API, ensuring its continued relevance and integration into evolving treatment protocols. The expanding healthcare infrastructure and improved diagnostic capabilities in various regions also contribute to the earlier and more accurate identification of fungal infections, leading to timely treatment initiation and thus sustaining API demand.
Despite its critical role, the 5-Fluocytosine API market is not without its hurdles and constraints. A significant challenge lies in the potential for the development of antifungal resistance. Over-reliance on any single antifungal agent can inadvertently lead to the emergence of resistant fungal strains, diminishing the efficacy of 5-Fluocytosine API over time and necessitating careful stewardship of its use. This poses a long-term threat to its market sustainability. Another restraint is the relatively narrow spectrum of activity compared to some newer generation antifungals, particularly against certain rare or highly resistant fungal pathogens. While effective against common culprits, its limitations in specific clinical scenarios can lead to the exploration of alternative treatments. Furthermore, the production of high-purity 5-Fluocytosine API can be complex and costly, impacting overall manufacturing economics and potentially leading to price sensitivities for certain market segments. Stringent regulatory requirements for API manufacturing and quality control also add to the operational expenses and can create barriers to entry for new players. The availability of alternative therapeutic options, including newer antifungal classes, while beneficial for patient care, can also fragment the market and present competitive pressures. Finally, the geographical concentration of manufacturing capabilities and the potential for supply chain disruptions due to geopolitical factors or unforeseen events can pose significant risks to market stability.
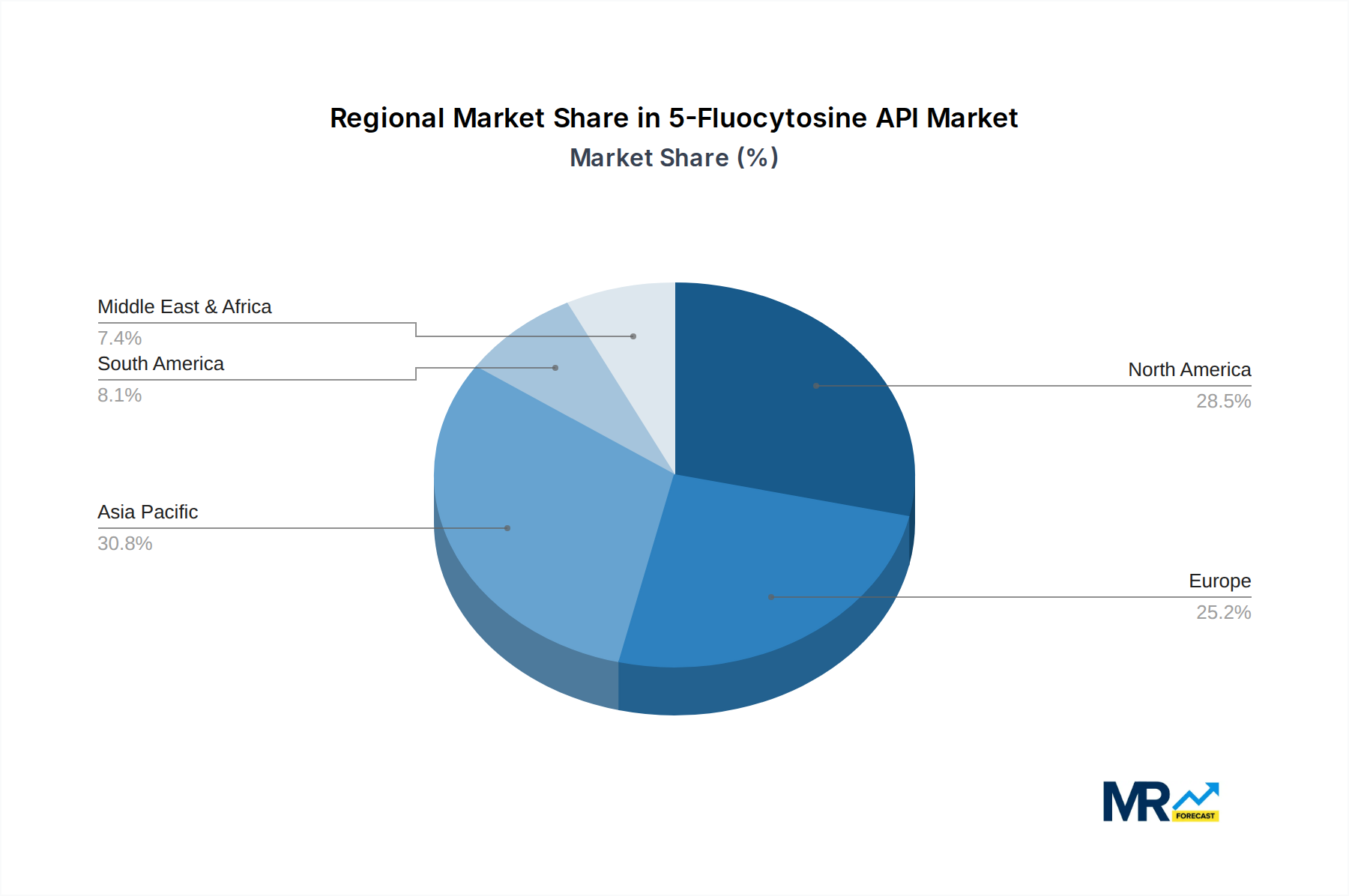
The 5-Fluocytosine API market is poised for significant dominance by specific regions and segments, driven by a complex interplay of healthcare infrastructure, disease prevalence, and market dynamics.
Key Dominating Segments:
Dominating Regions/Countries:
Several factors are acting as significant growth catalysts for the 5-Fluocytosine API industry. The persistent and, in some regions, increasing incidence of serious systemic fungal infections, particularly among immunocompromised individuals, remains a primary driver. Advancements in medical treatments leading to longer survival rates for patients with chronic diseases or undergoing complex procedures often result in a higher risk of opportunistic infections, thus sustaining demand. Furthermore, the ongoing research into combination therapies, where 5-Fluocytosine API's synergistic properties are being explored to enhance the efficacy and broaden the spectrum of antifungal treatments, opens up new avenues for its application and market penetration. The development and adoption of more sensitive diagnostic tools for fungal infections also contribute to earlier detection and treatment initiation, thereby boosting API consumption. Lastly, the strategic expansion of manufacturing capabilities by key players in regions with growing healthcare needs and cost-competitive production environments will serve as crucial growth catalysts, ensuring wider availability and accessibility of the API.
The 5-Fluocytosine API market is characterized by the presence of several key manufacturers and suppliers who are instrumental in its production and distribution. These companies are at the forefront of ensuring the availability of this essential antifungal API.
The 5-Fluocytosine API sector has witnessed several noteworthy developments over the study period, reflecting the industry's evolution and adaptation to market demands and technological advancements. These developments are crucial for understanding the trajectory of the market.
This comprehensive report on the 5-Fluocytosine API market provides an exhaustive overview of its dynamics, from historical performance to future projections. It meticulously analyzes the key market drivers, including the escalating burden of fungal infections and the critical role of 5-Fluocytosine API in treating immunocompromised patients. The report also addresses the inherent challenges and restraints, such as the potential for antifungal resistance and the complexities of high-purity API production. A detailed regional analysis identifies the dominant markets and their underlying reasons, while a segment-wise breakdown highlights the areas with the highest growth potential, particularly focusing on 5-Fluocytosine Tablet applications and 0.99 purity grades. Furthermore, the report identifies significant growth catalysts and provides an up-to-date list of leading players actively shaping the market. The inclusion of significant industry developments, presented chronologically, offers a valuable perspective on the sector's evolution. This report offers actionable intelligence for all stakeholders, enabling informed strategic decision-making within the dynamic 5-Fluocytosine API landscape.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 4.3% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 4.3%.
Key companies in the market include Jolly Healthcare, Manus Aktteva Biopharma LLP, VIVAN Life Sciences, Otto Chemie, national analytical corporation, Xinxiang Tuoxin Pharmaceutical Co.,Ltd., Wuhan Yinghe Pharmaceutical Co., Ltd., Hefei Home Sunshine Pharmaceutical, Hubei Maoerwo Biomedicine Co., Ltd., Hubei Widely Chemical Technology Co., Ltd., BESTPHARM CO,LTD., .
The market segments include Application, Type.
The market size is estimated to be USD 231.5 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "5-Fluocytosine API," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the 5-Fluocytosine API, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.